Sebelipase alfa for lysosomal acid lipase deficiency: 5-year treatment experience from a phase 2 open-label extension study
- PMID: 32657505
- PMCID: PMC7540377
- DOI: 10.1111/liv.14603
Sebelipase alfa for lysosomal acid lipase deficiency: 5-year treatment experience from a phase 2 open-label extension study
Abstract
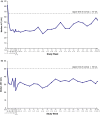
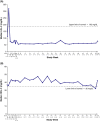
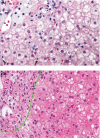
Background and aims: Lysosomal acid lipase deficiency is characterized by hepatomegaly and dyslipidaemia, which can lead to cirrhosis and premature atherosclerosis. Sebelipase alfa is an approved recombinant human lysosomal acid lipase. In an open-label extension study of adults with lysosomal acid lipase deficiency (LAL-CL04), sebelipase alfa treatment for 1 year reduced serum transaminase levels and liver fat content and improved serum lipid levels.
Methods: Final data from LAL-CL04 are reported herein for patients who received sebelipase alfa infusions (1.0 or 3.0 mg/kg every other week) for up to 5 years.
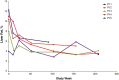
Results: Of 8 patients enrolled, 7 received sebelipase alfa for 224-260 weeks; 1 was lost to follow-up. Median baseline levels of alanine aminotransferase and aspartate aminotransferase (81.5 and 50.0 U/L, respectively) were decreased through the end-of-study visit (54.0 and 34.0 U/L). Median low-density lipoprotein cholesterol decreased from 113 to 78 mg/dL, total cholesterol decreased from 171 to 132 mg/dL, and high-density lipoprotein cholesterol increased from 37 to 42 mg/dL. Most treatment-emergent adverse events were nonserious (99%), mild/moderate (98%) and unrelated to sebelipase alfa (87%); no patient discontinued as a result of treatment-emergent adverse events. One patient had 2 serious treatment-emergent adverse events (cholecystitis and cholelithiasis; assessed as unlikely related to sebelipase alfa). Two patients had 20 nonserious infusion-associated reactions in weeks 6-38; all were manageable. One patient tested positive for antidrug antibodies (single occurrence).
Conclusions: Sebelipase alfa was well tolerated and improved serum transaminase and lipid levels for up to 5 years in adults with lysosomal acid lipase deficiency.
Trial registration number: ClinicalTrials.gov record NCT01488097.
Keywords: enzyme replacement therapy; lipids; liver; lysosomal storage diseases; transaminases.
© 2020 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd.
Conflict of interest statement
VM received funding for participation as a study investigator and an honorarium for chairing an educational session from Alexion Pharmaceuticals, Inc JBA and RS have received honoraria for educational sessions and funding from Alexion Pharmaceuticals, Inc for participation as study investigators. JK has given lectures on this topic supported by Alexion Pharmaceuticals, Inc, and has received funding from Alexion Pharmaceuticals, Inc for participation as a study investigator. MB is a member of the LALD scientific registry and has received honoraria for participation in advisory boards and funding from Alexion Pharmaceuticals, Inc for participation as a study investigator. CBW received consultancy fees and funding from Alexion Pharmaceuticals, Inc for providing education on lysosomal diseases. SM and FA are employees of and may own stock/options in Alexion Pharmaceuticals, Inc
Figures

Similar articles
-
Sebelipase alfa over 52 weeks reduces serum transaminases, liver volume and improves serum lipids in patients with lysosomal acid lipase deficiency.J Hepatol. 2014 Nov;61(5):1135-42. doi: 10.1016/j.jhep.2014.06.022. Epub 2014 Jun 30. J Hepatol. 2014. PMID: 24993530 Free PMC article. Clinical Trial.
-
Sebelipase alfa in children and adults with lysosomal acid lipase deficiency: Final results of the ARISE study.J Hepatol. 2022 Mar;76(3):577-587. doi: 10.1016/j.jhep.2021.10.026. Epub 2021 Nov 10. J Hepatol. 2022. PMID: 34774639
-
Long-term survival with sebelipase alfa enzyme replacement therapy in infants with rapidly progressive lysosomal acid lipase deficiency: final results from 2 open-label studies.Orphanet J Rare Dis. 2021 Jan 6;16(1):13. doi: 10.1186/s13023-020-01577-4. Orphanet J Rare Dis. 2021. PMID: 33407676 Free PMC article.
-
Enzyme replacement therapy in lysosomal acid lipase deficiency (LAL-D): a systematic literature review.Ther Adv Rare Dis. 2021 Jul 18;2:26330040211026928. doi: 10.1177/26330040211026928. eCollection 2021 Jan-Dec. Ther Adv Rare Dis. 2021. PMID: 37181111 Free PMC article. Review.
-
Sebelipase Alfa: A Review in Lysosomal Acid Lipase Deficiency.Am J Cardiovasc Drugs. 2016 Dec;16(6):461-468. doi: 10.1007/s40256-016-0203-2. Am J Cardiovasc Drugs. 2016. PMID: 27878737 Review.
Cited by
-
Circulating miRNAs as Biomarkers for Mitochondrial Neuro-Gastrointestinal Encephalomyopathy.Int J Mol Sci. 2021 Apr 1;22(7):3681. doi: 10.3390/ijms22073681. Int J Mol Sci. 2021. PMID: 33916195 Free PMC article.
-
Genetics Is of the Essence to Face NAFLD.Biomedicines. 2021 Sep 30;9(10):1359. doi: 10.3390/biomedicines9101359. Biomedicines. 2021. PMID: 34680476 Free PMC article. Review.
-
Lessons Learned From the Long-Term Use of Enzyme Replacement Therapy in the Treatment of Lysosomal Acid Lipase Deficiency.J Pediatr Gastroenterol Nutr. 2022 Jun 1;74(6):726-727. doi: 10.1097/MPG.0000000000003453. Epub 2022 May 17. J Pediatr Gastroenterol Nutr. 2022. PMID: 35576532 Free PMC article. No abstract available.
-
Lanifibranor Reduces Inflammation and Improves Dyslipidemia in Lysosomal Acid Lipase-Deficient Mice.Gastro Hep Adv. 2024 May 28;3(6):711-723. doi: 10.1016/j.gastha.2024.05.006. eCollection 2024. Gastro Hep Adv. 2024. PMID: 39280921 Free PMC article.
-
Recent insights into lysosomal acid lipase deficiency.Trends Mol Med. 2023 Jun;29(6):425-438. doi: 10.1016/j.molmed.2023.03.001. Epub 2023 Apr 5. Trends Mol Med. 2023. PMID: 37028992 Free PMC article. Review.
References
-
- Grabowski GA, Charnas L, Du H. Lysosomal acid lipase deficiencies: the Wolman disease/cholesteryl ester storage disease spectrum In: Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Antonarakis SE, Ballabio A, eds. Metabolic and Molecular Bases of Inherited Disease, 8th ed New York, NY: McGraw‐Hill; 2012.
-
- Rader DJ. Lysosomal acid lipase deficiency ‐ a new therapy for a genetic lipid disease. N Engl J Med. 2015;373(11):1071‐1073. - PubMed
-
- Reiner Ž, Guardamagna O, Nair D, et al. Lysosomal acid lipase deficiency–an under‐recognized cause of dyslipidaemia and liver dysfunction. Atherosclerosis. 2014;235(1):21‐30. - PubMed
-
- Bernstein DL, Hulkova H, Bialer MG, Desnick RJ. Cholesteryl ester storage disease: review of the findings in 135 reported patients with an underdiagnosed disease. J Hepatol. 2013;58(6):1230‐1243. - PubMed
-
- Hamilton J, Jones I, Srivastava R, Galloway P. A new method for the measurement of lysosomal acid lipase in dried blood spots using the inhibitor Lalistat 2. Clin Chim Acta. 2012;413(15–16):1207‐1210. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources

