Fibrous Dysplasia/McCune-Albright Syndrome: A Rare, Mosaic Disease of Gα s Activation
- PMID: 31673695
- PMCID: PMC7127130
- DOI: 10.1210/endrev/bnz011
Fibrous Dysplasia/McCune-Albright Syndrome: A Rare, Mosaic Disease of Gα s Activation
Abstract
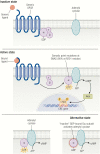
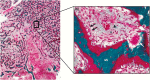
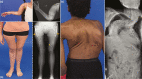
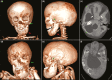
Fibrous dysplasia/McCune-Albright syndrome (FD/MAS) is a rare disorder of striking complexity. It arises from somatic, gain-of-function mutations in GNAS, leading to mosaic Gα s activation and inappropriate production of intracellular cyclic adenosine monophosphate (cAMP). The clinical phenotype is largely determined by the location and extent of affected tissues, and the pathophysiological effects of Gα s activation within these tissues. In bone, Gα s activation results in impaired differentiation of skeletal stem cells, leading to discrete skeletal lesions prone to fracture, deformity, and pain. Extraskeletal manifestations include a variable combination of hyperpigmented macules and hyperfunctioning endocrinopathies. Distinctive age-related changes in disease development has key effects on histologic, radiographic, and clinical features. FD/MAS thus presents along a uniquely broad clinical spectrum, and the resulting challenges in diagnosis and management can be difficult for clinicians. This review presents FD/MAS in the context of a mosaic disorder of Gα s activation, providing an intellectual framework within which to understand, evaluate, and treat this interesting disease. It includes a comprehensive summary of current understanding of FD/MAS pathogenesis, and a detailed discussion of clinical presentation and management. Critical areas of unmet need are highlighted, including discussion of key challenges and potential solutions to advance research and clinical care in FD/MAS.
Keywords: fibroblast growth factor 23; growth hormone excess; metabolic bone disease; precocious puberty; skeletal stem cells; somatic mosaicism.
Published by Oxford University Press on behalf of the Endocrine Society 2019.
Figures

Similar articles
-
The Clinical Spectrum of McCune-Albright Syndrome and Its Management.Horm Res Paediatr. 2019;92(6):347-356. doi: 10.1159/000504802. Epub 2019 Dec 19. Horm Res Paediatr. 2019. PMID: 31865341 Free PMC article. Review.
-
Fibrous Dysplasia/McCune-Albright Syndrome: Clinical and Translational Perspectives.Curr Osteoporos Rep. 2016 Oct;14(5):178-86. doi: 10.1007/s11914-016-0317-0. Curr Osteoporos Rep. 2016. PMID: 27492469 Free PMC article. Review.
-
Extent of Extraskeletal Manifestations of Fibrous Dysplasia/McCune-Albright Syndrome in Patients with Mazabraud's Syndrome.Calcif Tissue Int. 2022 Mar;110(3):334-340. doi: 10.1007/s00223-021-00918-0. Epub 2021 Dec 2. Calcif Tissue Int. 2022. PMID: 34854944
-
Brain and eye involvement in McCune-Albright Syndrome: clinical and translational insights.Front Endocrinol (Lausanne). 2023 May 19;14:1092252. doi: 10.3389/fendo.2023.1092252. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37274327 Free PMC article. Review.
-
Choosing the Best Tissue and Technique to Detect Mosaicism in Fibrous Dysplasia/McCune-Albright Syndrome (FD/MAS).Genes (Basel). 2024 Jan 18;15(1):120. doi: 10.3390/genes15010120. Genes (Basel). 2024. PMID: 38255009 Free PMC article.
Cited by
-
Serum Phosphorus as a Driver of Skeletal Morbidity in Fibrous Dysplasia.J Clin Endocrinol Metab. 2024 Apr 19;109(5):1334-1340. doi: 10.1210/clinem/dgad671. J Clin Endocrinol Metab. 2024. PMID: 37975816 Free PMC article.
-
Cell surface-bound La protein regulates the cell fusion stage of osteoclastogenesis.Nat Commun. 2023 Feb 4;14(1):616. doi: 10.1038/s41467-023-36168-x. Nat Commun. 2023. PMID: 36739273 Free PMC article.
-
Molecular basis of normal and pathological puberty: from basic mechanisms to clinical implications.Lancet Diabetes Endocrinol. 2023 Mar;11(3):203-216. doi: 10.1016/S2213-8587(22)00339-4. Epub 2023 Jan 5. Lancet Diabetes Endocrinol. 2023. PMID: 36620967 Free PMC article. Review.
-
Phenotyping Pain in Patients With Fibrous Dysplasia/McCune-Albright Syndrome.J Clin Endocrinol Metab. 2024 Feb 20;109(3):771-782. doi: 10.1210/clinem/dgad589. J Clin Endocrinol Metab. 2024. PMID: 37804088 Free PMC article.
-
Safety of therapy with and withdrawal from denosumab in fibrous dysplasia and McCune-Albright syndrome: an observational study.J Bone Miner Res. 2021 Sep;36(9):1729-1738. doi: 10.1002/jbmr.4380. Epub 2021 Jun 10. J Bone Miner Res. 2021. PMID: 34076303 Free PMC article.
References
-
- Boyce AM, Florenzano P, de Castro LF, Collins MT. Fibrous dysplasia/McCune-Albright syndrome. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, Amemiya A, eds. GeneReviews®. Seattle, WA: University of Washington, Seattle; 1993-2020. - PubMed
-
- Weinstein LS, Shenker A, Gejman PV, Merino MJ, Friedman E, Spiegel AM. Activating mutations of the stimulatory G protein in the McCune-Albright syndrome. N Engl J Med. 1991;325(24):1688–1695. - PubMed
-
- Riminucci M, Fisher LW, Majolagbe A, et al. . A novel GNAS1 mutation, R201G, in McCune-Albright syndrome. J Bone Miner Res. 1999;14(11):1987–1989. - PubMed
-
- Idowu BD, Al-Adnani M, O’Donnell P, et al. . A sensitive mutation-specific screening technique for GNAS1 mutations in cases of fibrous dysplasia: the first report of a codon 227 mutation in bone. Histopathology. 2007;50(6):691–704. - PubMed

