Acute physiological changes caused by complement activators and amphotericin B-containing liposomes in mice
- PMID: 30880965
- PMCID: PMC6396670
- DOI: 10.2147/IJN.S187139
Acute physiological changes caused by complement activators and amphotericin B-containing liposomes in mice
Abstract
Purpose: Undesirable complement (C) activation by nanomedicines can entail an adverse immune reaction known as C activation-related pseudoallergy (CARPA) in sensitive patients. The syndrome includes cardiopulmonary, hemodynamic, and a variety of other physiological changes that have been well described in man, pigs, dogs, and rats. However, the information on CARPA is scarce and ambiguous in mice, a species widely used in preclinical studies. The present study aimed to fill this gap by exploring signs of CARPA in mice following i.v. administration of AmBisome and Abelcet, which are nano-formulations of Amphotericin B with high risk to cause CARPA.
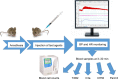
Materials and methods: Anesthetized NMRI mice were intravenously injected with liposomal amphotericin B (Abelcet and AmBisome; 30-300 mg phospholipid/kg), drug-free high cholesterol multilamellar vesicles (HC-MLV), and positive controls, cobra venom factor (CVF) and zymosan, followed by the measurement of blood pressure (BP), heart rate, white blood cell, and platelet counts and plasma thromboxane B2 (TXB2) levels. C activation was assessed by C3a ELISA, a C3 consumption assay (PAN-C3) and a modified sheep red blood cell hemolytic assay.
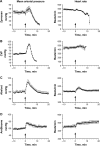
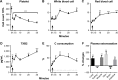
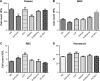
Results: All test agents, except HC-MLV, caused transient hypertension, thrombocytopenia, and elevation of plasma TXB2, which were paralleled by significant rises of plasma C3a in CVF and zymosan-treated animals, wherein the initial hypertension turned into hypotension and shock. Abelcet and AmBisome caused minor, delayed rise of C3a that was not associated with hypertension. The C3a receptor inhibitor SB-290157 attenuated the hypertension caused by Abelcet and decreased the BP thereafter.
Conclusion: The parallelism between C3a anaphylatoxin production and severity of physiological changes caused by the different agents is consistent with CARPA underlying these changes. Although the reactive dose of liposomal phospholipids was substantially higher than that in other species (pigs, dogs), the mouse seems suitable for studying the mechanism of hypersensitivity reactions to liposomal formulations of amphotericin B, a frequent side effect of these drugs.
Keywords: TXB2; anaphylatoxins; cholesterol; cobra venom factor; hypersensitivity; infusion reactions; platelets; zymosan.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures


Similar articles
-
Complement Activation-Related Pathophysiological Changes in Anesthetized Rats: Activator-Dependent Variations of Symptoms and Mediators of Pseudoallergy.Molecules. 2019 Sep 9;24(18):3283. doi: 10.3390/molecules24183283. Molecules. 2019. PMID: 31505853 Free PMC article.
-
Features of complement activation-related pseudoallergy to liposomes with different surface charge and PEGylation: comparison of the porcine and rat responses.J Control Release. 2014 Dec 10;195:2-10. doi: 10.1016/j.jconrel.2014.08.009. Epub 2014 Aug 19. J Control Release. 2014. PMID: 25148822
-
The Hypertensive Effect of Amphotericin B-Containing Liposomes (Abelcet) in Mice: Dissecting the Roles of C3a and C5a Anaphylatoxins, Macrophages and Thromboxane.Biomedicines. 2022 Jul 21;10(7):1764. doi: 10.3390/biomedicines10071764. Biomedicines. 2022. PMID: 35885068 Free PMC article.
-
A porcine model of complement-mediated infusion reactions to drug carrier nanosystems and other medicines.Adv Drug Deliv Rev. 2012 Dec;64(15):1706-16. doi: 10.1016/j.addr.2012.07.005. Epub 2012 Jul 20. Adv Drug Deliv Rev. 2012. PMID: 22820530 Review.
-
Complement activation as a bioequivalence issue relevant to the development of generic liposomes and other nanoparticulate drugs.Biochem Biophys Res Commun. 2015 Dec 18;468(3):490-7. doi: 10.1016/j.bbrc.2015.06.177. Epub 2015 Jul 14. Biochem Biophys Res Commun. 2015. PMID: 26182876 Review.
Cited by
-
Targeting vascular inflammation through emerging methods and drug carriers.Adv Drug Deliv Rev. 2022 May;184:114180. doi: 10.1016/j.addr.2022.114180. Epub 2022 Mar 7. Adv Drug Deliv Rev. 2022. PMID: 35271986 Free PMC article. Review.
-
The Critical Choice of Animal Models in Nanomedicine Safety Assessment: A Lesson Learned From Hemoglobin-Based Oxygen Carriers.Front Immunol. 2020 Oct 26;11:584966. doi: 10.3389/fimmu.2020.584966. eCollection 2020. Front Immunol. 2020. PMID: 33193403 Free PMC article. Review.
-
Comirnaty-induced cardiopulmonary distress and other symptoms of complement-mediated pseudo-anaphylaxis in a hyperimmune pig model: Causal role of anti-PEG antibodies.Vaccine X. 2024 May 23;19:100497. doi: 10.1016/j.jvacx.2024.100497. eCollection 2024 Aug. Vaccine X. 2024. PMID: 38933697 Free PMC article.
-
The "C3aR Antagonist" SB290157 is a Partial C5aR2 Agonist.Front Pharmacol. 2021 Jan 21;11:591398. doi: 10.3389/fphar.2020.591398. eCollection 2020. Front Pharmacol. 2021. PMID: 33551801 Free PMC article.
-
siRNA-mediated reduction of a circulating protein in swine using lipid nanoparticles.Mol Ther Methods Clin Dev. 2024 Apr 27;32(2):101258. doi: 10.1016/j.omtm.2024.101258. eCollection 2024 Jun 13. Mol Ther Methods Clin Dev. 2024. PMID: 38779336 Free PMC article.
References
-
- Szebeni J. Complement activation-related pseudoallergy: a new class of drug-induced acute immune toxicity. Toxicology. 2005;216(2–3):106–121. - PubMed
-
- Szebeni J. Complement activation-related pseudoallergy: a stress reaction in blood triggered by nanomedicines and biologicals. Mol Immunol. 2014;61(2):163–173. - PubMed
-
- Dézsi L, Rosivall L, Hamar P, Szebeni J, Szénási G. Rodent models of complement activation-related pseudoallergy: inducers, symptoms, inhibitors and reaction mechanisms. Eur J Nanomed. 2015;7(1):15.
-
- Proctor LM, Moore TA, Monk PN, Sanderson SD, Taylor SM, Woodruff TM. Complement factors C3a and C5a have distinct hemodynamic effects in the rat. Int Immunopharmacol. 2009;9(6):800–806. - PubMed
-
- Engström G, Hedblad B, Berglund G, Janzon L, Lindgärde F. Plasma levels of complement C3 is associated with development of hypertension: a longitudinal cohort study. J Hum Hypertens. 2007;21(4):276–282. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

