Paracetamol versus placebo for knee and hip osteoarthritis
- PMID: 30801133
- PMCID: PMC6388567
- DOI: 10.1002/14651858.CD013273
Paracetamol versus placebo for knee and hip osteoarthritis
Abstract
Background: Paracetamol (acetaminophen) is vastly recommended as the first-line analgesic for osteoarthritis of the hip or knee. However, there has been controversy about this recommendation given recent studies have revealed small effects of paracetamol when compared with placebo. Nonetheless, past studies have not systematically reviewed and appraised the literature to investigate the effects of this drug on specific osteoarthritis sites, that is, hip or knee, or on the dose used.
Objectives: To assess the benefits and harms of paracetamol compared with placebo in the treatment of osteoarthritis of the hip or knee.
Search methods: We searched the Cochrane Central Register of Controlled Trials, MEDLINE, Embase, AMED, CINAHL, Web of Science, LILACS, and International Pharmaceutical Abstracts to 3 October 2017, and ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform (ICTRP) portal on 20 October 2017.
Selection criteria: We included randomised controlled trials comparing paracetamol with placebo in adults with osteoarthritis of the hip or knee. Major outcomes were pain, function, quality of life, adverse events and withdrawals due to adverse events, serious adverse events, and abnormal liver function tests.
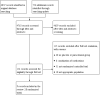
Data collection and analysis: Two review authors used standard Cochrane methods to collect data, and assess risk of bias and quality of the evidence. For pooling purposes, we converted pain and physical function (Western Ontario and McMaster Universities Osteoarthritis Index function) scores to a common 0 (no pain or disability) to 100 (worst possible pain or disability) scale.
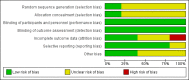
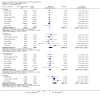
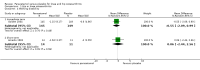
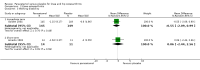
Main results: We identified 10 randomised placebo-controlled trials involving 3541 participants with hip or knee osteoarthritis. The paracetamol dose varied from 1.95 g/day to 4 g/day, and the majority of trials followed participants for three months only. Most trials did not clearly report randomisation and concealment methods and were at unclear risk of selection bias. Trials were at low risk of performance, detection, and reporting bias.At 3 weeks' to 3 months' follow-up, there was high-quality evidence that paracetamol provided no clinically important improvements in pain and physical function. Mean reduction in pain was 23 points (0 to 100 scale, lower scores indicated less pain) with placebo and 3.23 points better (5.43 better to 1.02 better) with paracetamol, an absolute reduction of 3% (1% better to 5% better, minimal clinical important difference 9%) and relative reduction of 5% (2% better to 8% better) (seven trials, 2355 participants). Physical function improved by 12 points on a 0 to 100 scale (lower scores indicated better function) with placebo and was 2.9 points better (0.95 better to 4.89 better) with paracetamol, an absolute improvement of 3% (1% better to 5% better, minimal clinical important difference 10%) and relative improvement of 5% (2% better to 9% better) (7 trials, 2354 participants).High-quality evidence from eight trials indicated that the incidence of adverse events was similar between groups: 515/1586 (325 per 1000) in the placebo group versus 537/1666 (328 per 1000, range 299 to 360) in the paracetamol group (risk ratio (RR) 1.01, 95% confidence interval (CI) 0.92 to 1.11). There was less certainty (moderate-quality evidence) around the risk of serious adverse events, withdrawals due to adverse events, and the rate of abnormal liver function tests, due to wide CIs or small event rates, indicating imprecision. Seventeen of 1480 (11 per 1000) people treated with placebo and 28/1729 (16 per 1000, range 8 to 29) people treated with paracetamol experienced serious adverse events (RR 1.36, 95% CI 0.73 to 2.53; 6 trials). The incidence of withdrawals due to adverse events was 65/1000 participants in with placebo and 77/1000 (range 59 to 100) participants with paracetamol (RR 1.19, 95% CI 0.91 to 1.55; 7 trials). Abnormal liver function occurred in 18/1000 participants treated with placebo and 70/1000 participants treated with paracetamol (RR 3.79, 95% CI 1.94 to 7.39), but the clinical importance of this effect was uncertain. None of the trials reported quality of life.Subgroup analyses indicated that the effects of paracetamol on pain and function did not differ according to the dose of paracetamol (3.0 g/day or less versus 3.9 g/day or greater).
Authors' conclusions: Based on high-quality evidence this review confirms that paracetamol provides only minimal improvements in pain and function for people with hip or knee osteoarthritis, with no increased risk of adverse events overall. Subgroup analysis indicates that the effects on pain and function do not differ according to the dose of paracetamol. Due to the small number of events, we are less certain if paracetamol use increases the risk of serious adverse events, withdrawals due to adverse events, and rate of abnormal liver function tests.Current clinical guidelines consistently recommend paracetamol as the first-line analgesic medication for hip or knee osteoarthritis, given its low absolute frequency of substantive harm. However, our results call for reconsideration of these recommendations.
Conflict of interest statement
AOL: no conflict of interest.
GCM, MBP, PHF, ROD, AJM, and MLF: conducted a systematic review that investigated the benefits of paracetamol for patients with osteoarthritis and spinal pain published in the
ROD and AJM: investigators on the PACE (Placebo, Acetaminophen (paracetamol) or Celecoxib Efficacy Studies) trial of paracetamol in low back pain which was jointly funded by the National Health and Medical Research Council of Australia and GlaxoSmithKline Australia (a manufacturer of paracetamol).
AM: received research funding support from GlaxoSmithKline for a PhD scholarship.
DJH is a consultant for Flexion, Merck Serono, and Nestlé.
Figures














Comment in
-
Paracetamol as first line for treatment of knee and hip osteoarthritis.BMJ Evid Based Med. 2020 Feb;25(1):40. doi: 10.1136/bmjebm-2019-111213. Epub 2019 Jun 15. BMJ Evid Based Med. 2020. PMID: 31203255 Review. No abstract available.
-
Is paracetamol better than placebo for knee and hip osteoarthritis? A Cochrane review summary with commentary.Int J Rheum Dis. 2020 Apr;23(4):595-596. doi: 10.1111/1756-185X.13827. Int J Rheum Dis. 2020. PMID: 32286015 No abstract available.
Similar articles
-
Chondroitin for osteoarthritis.Cochrane Database Syst Rev. 2015 Jan 28;1(1):CD005614. doi: 10.1002/14651858.CD005614.pub2. Cochrane Database Syst Rev. 2015. PMID: 25629804 Free PMC article. Review.
-
Tramadol for osteoarthritis.Cochrane Database Syst Rev. 2019 May 27;5(5):CD005522. doi: 10.1002/14651858.CD005522.pub3. Cochrane Database Syst Rev. 2019. PMID: 31132298 Free PMC article.
-
Celecoxib for osteoarthritis.Cochrane Database Syst Rev. 2017 May 22;5(5):CD009865. doi: 10.1002/14651858.CD009865.pub2. Cochrane Database Syst Rev. 2017. PMID: 28530031 Free PMC article. Review.
-
Efficacy and safety of paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials.BMJ. 2015 Mar 31;350:h1225. doi: 10.1136/bmj.h1225. BMJ. 2015. PMID: 25828856 Free PMC article. Review.
-
Adjunctive therapies in addition to land-based exercise therapy for osteoarthritis of the hip or knee.Cochrane Database Syst Rev. 2022 Oct 17;10(10):CD011915. doi: 10.1002/14651858.CD011915.pub2. Cochrane Database Syst Rev. 2022. PMID: 36250418 Free PMC article. Review.
Cited by
-
Patterns of pain medication usage and self-reported pain in older Irish adults with osteoarthritis: A latent class analysis of data from the Irish Longitudinal Study on Ageing.BMC Musculoskelet Disord. 2024 Oct 2;25(1):773. doi: 10.1186/s12891-024-07854-8. BMC Musculoskelet Disord. 2024. PMID: 39358713 Free PMC article.
-
Oral non-steroidal anti-inflammatory drugs versus other oral analgesic agents for acute soft tissue injury.Cochrane Database Syst Rev. 2020 Aug 12;8(8):CD007789. doi: 10.1002/14651858.CD007789.pub3. Cochrane Database Syst Rev. 2020. PMID: 32797734 Free PMC article.
-
New Trends in Pharmacological Treatments for Osteoarthritis.Front Pharmacol. 2021 Apr 15;12:645842. doi: 10.3389/fphar.2021.645842. eCollection 2021. Front Pharmacol. 2021. PMID: 33935742 Free PMC article. Review.
-
Nortriptyline for pain in knee osteoarthritis: a double-blind randomised controlled trial in New Zealand general practice.Br J Gen Pract. 2021 Jun 24;71(708):e538-e546. doi: 10.3399/BJGP.2020.0797. Print 2021 Jul. Br J Gen Pract. 2021. PMID: 33571950 Free PMC article. Clinical Trial.
-
PEER umbrella systematic review of systematic reviews: Management of osteoarthritis in primary care.Can Fam Physician. 2020 Mar;66(3):e89-e98. Can Fam Physician. 2020. PMID: 32165479 Free PMC article.
References
References to studies included in this review
Altman 2007 {published data only}
-
- Altman RD, Zinsenheim JR, Temple AR, Schweinle JE. Three‐month efficacy and safety of acetaminophen extended‐release for osteoarthritis pain of the hip or knee: a randomized, double‐blind, placebo‐controlled study. Osteoarthritis and Cartilage 2007;15:454‐61. - PubMed
Amadio 1983 {published data only}
-
- Amadio P, Doyle M. Evaluation of acetaminophen in the management of osteoarthritis of the knee. Current Therapeutic Research – Clinical and Experimental 1983;34(1):59‐66.
Case 2003 {published data only}
-
- Case JP, Baliunas AJ, Block JA. Lack of efficacy of acetaminophen in treating symptomatic knee osteoarthritis: a randomized, double‐blind, placebo‐controlled comparison trial with diclofenac sodium. Archives of Internal Medicine 2003;163(2):169‐78. - PubMed
Golden 2004 {published data only}
-
- Golden HE, Moskowitz RW, Minic M. Analgesic efficacy and safety of nonprescription doses of naproxen sodium compared with acetaminophen in the treatment of osteoarthritis of the knee. American Journal of Therapeutics 2004;11:85‐94. - PubMed
Herrero‐Beaumont 2007 {published data only}
-
- Herrero‐Beaumont G, Ivorra JA, Carmen Trabado M, Blanco FJ, Benito P, Martín‐Mola E, et al. Glucosamine sulfate in the treatment of knee osteoarthritis symptoms: a randomized, double‐blind, placebo‐controlled study using acetaminophen as a side comparator. Arthritis and Rheumatism 2007;56(2):555‐67. - PubMed
Miceli‐Richard 2004 {published data only}
Pincus 2004a {published data only}
-
- Pincus T, Koch G, Lei H, Mangal B, Mangal B, Sokka T, Moskowitz R, et al. Patient Preference for Placebo, Acetaminophen (paracetamol) or Celecoxib Efficacy Studies (PACES): two randomised, double blind, placebo controlled, crossover clinical trials in patients with knee or hip osteoarthritis. Annals of the Rheumatic Diseases 2004;63(8):931‐9. - PMC - PubMed
Pincus 2004b {published data only}
-
- Pincus T, Koch G, Lei H, Mangal B, Mangal B, Sokka T, Moskowitz R, et al. Patient Preference for Placebo, Acetaminophen (paracetamol) or Celecoxib Efficacy Studies (PACES): two randomised, double blind, placebo controlled, crossover clinical trials in patients with knee or hip osteoarthritis. Annals of the Rheumatic Diseases 2004;63(8):931‐9. - PMC - PubMed
Prior 2014 {published data only}
-
- Prior MJ, Harrison DD, Frustaci ME. A randomized, double‐blind, placebo‐controlled 12 week trial of acetaminophen extended release for the treatment of signs and symptoms of osteoarthritis. Current Medical Research & Opinion 2014;30(11):2377‐87. - PubMed
Zoppi 1995 {published data only}
-
- Zoppi M, Peretti G, Boccard E. Placebo‐controlled study of the analgesic efficacy of an effervescent formulation of 500 mg paracetamol in arthritis of the knee or the hip. European Journal of Pain 1995;16:1‐2.
References to studies excluded from this review
Ha 2016 {published data only}
-
- Ha CW, Park YB, Min BW, Han SB, Lee JH, Won YY, et al. Prospective, randomized, double‐blinded, double‐dummy and multicenter phase IV clinical study comparing the efficacy and safety of PG201 (Layla) and SKI306X in patients with osteoarthritis. Journal of Ethnopharmacology 2016;181:1‐7. - PubMed
Lao 2015 {published data only}
-
- Lao L, Hochberg M, Lee DY, Gilpin AM, Fong HH, Langenberg P, et al. Huo‐Luo‐Xiao‐Ling (HLXL)‐Dan, a Traditional Chinese Medicine, for patients with osteoarthritis of the knee: a multi‐site, randomized, double‐blind, placebo‐controlled phase II clinical trial. Osteoarthritis and Cartilage 2015;23(12):2102‐8. - PMC - PubMed
Papou 2015 {published data only}
-
- Papou A, Hussain S, McWilliams D, Zhang W, Doherty M. Sensitivity to change of SF‐36 health survey and patient generated index in people with chronic knee pain commenced on oral analgesia: analysis of data from a clinical trial. Annals of the Rheumatic Diseases 2015;74:363. - PubMed
Park 2015 {published data only}
-
- Park YB, Ha CW, Cho SD, Lee MC, Lee JH, Seo SS, et al. A randomized study to compare the efficacy and safety of extended‐release and immediate‐release tramadol HCl/acetaminophen in patients with acute pain following total knee replacement. Current Medical Research and Opinion 2015;31(1):75‐84. - PubMed
Skou 2015 {published data only}
-
- Skou ST, Rasmussen S, Laursen MB, Rathleff MS, Arendt‐Nielsen L, Simonsen O, et al. The efficacy of 12 weeks non‐surgical treatment for patients not eligible for total knee replacement: a randomized controlled trial with 1‐year follow‐up. Osteoarthritis and Cartilage 2015;23(9):1465‐75. - PubMed
van Tunen 2016 {published data only}
-
- Tunen JA, Leeden M, Bos WH, Cheung J, Esch M, Gerritsen M, et al. Optimization of analgesics for greater exercise therapy participation among patients with knee osteoarthritis and severe pain: a feasibility study. Arthritis Care and Research 2016;68(3):332‐40. - PubMed
Verkleij 2015 {published data only}
Zheng 2015 {published data only}
-
- Zheng Y, Kostenbader K, Barrett T, Hisaw E, Giuliani MJ, Chen Y, Young JL. Tolerability of biphasic‐release hydrocodone bitartrate/acetaminophen tablets (MNK‐155): a phase III, multicenter, open‐label study in patients with osteoarthritis or chronic low back pain. Clinical Therapeutics 2015;1(37):1235‐47. - PubMed
References to ongoing studies
ACTRN12613000840785 {published data only}
-
- A randomized, blinded, comparator‐controlled trial investigating a 28‐day course of Lyrica in participants with knee osteoarthritis who exhibit neuropathic pain, compared with a 28‐day course of acetaminophen. Ongoing study 17 June 2013.
NCT01105936 {published data only}
-
- A randomised, double‐blind, evaluation of the effects of paracetamol on the BOLD fMRI response to painful stimuli in subjects with osteoarthritis. Ongoing study 1 September 2010.
NCT01420666 {published data only}
-
- Maxi‐Analgesic OA Study: multicentre, double‐blind, placebo‐controlled, randomized, parallel group comparison of the effects of Maxigesic 325 with acetaminophen or ibuprofen on patients with pain from osteoarthritis. Ongoing study 22 August 2011.
NCT02311881 {published data only}
-
- An efficacy and safety study of sustained‐release paracetamol in subjects with osteoarthritis. Ongoing study 9 December 2014.
NCT02845271 {published data only}
-
- Proof‐of‐concept study to assess the efficacy, tolerability and safety of a single intraarticular dose of GZ389988 versus placebo in patients with painful osteoarthritis of the knee. Ongoing study 27 July 2016.
Additional references
Altman 1996
-
- Altman R, Brandt K, Hochberg M, Moskowitz R, Bellamy N, Bloch DA, et al. Design and conduct of clinical trials in patients with osteoarthritis: recommendations from a task force of the Osteoarthritis Research Society. Results from a workshop. Osteoarthritis Cartilage 1996;4(4):217‐43. - PubMed
Bartels 2016
Barten 2015
Barthel 2010
Basedow 2015
Brand 2008
-
- Brand CA. The role of self‐management in designing care for people with osteoarthritis of the hip and knee. Medical Journal of Australia 2008;189(S):25‐8. - PubMed
Brand 2014
-
- Brand CA, Harrison C, Tropea J, Hinman RS, Britt H, Bennell K. Management of osteoarthritis in general practice in Australia. Arthritis Care and Research 2014;66:551‐8. - PubMed
Buckwalter 2006
-
- Buckwalter JA, Martin JA. Osteoarthritis. Advanced Drug Delivery Reviews 2006;20(58):150‐67. - PubMed
Crofford 2001
-
- Crofford LJ. Rational use of analgesic and antiinflammatory drugs. New England Journal of Medicine 2001;20(345):1844‐6. - PubMed
Cross 2014
-
- Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M, et al. The global burden of hip and knee osteoarthritis: estimates from the Global Burden of Disease 2010 study. Annals of the Rheumatic Diseases 2014;73:1323‐30. - PubMed
Derry 2016
Egger 1997
-
- Egger M, Zellweger‐Zähner T, Schneider M, Junker C, Lengeler C, Antes G. Language bias in randomised controlled trials published in English and German. Lancet 1997;2(305):326‐9. - PubMed
Egger 2003
-
- Egger M, Jüni P, Bartlett C, Holenstein F, Sterne J. How important are comprehensive literature searches and the assessment of trial quality in systematic reviews? Empirical study. Health Technology Assessment (Winchester, England) 2003;7(1):1‐76. - PubMed
Ghogomu 2014
-
- Ghogomu EA, Maxwell LJ, Buchbinder R, Rader T, Pardo Pardo J, Johnston RV, et al. Updated method guidelines for Cochrane musculoskeletal group systematic reviews and metaanalyses. Journal of Rheumatology 2014;41(2):194‐205. - PubMed
Graham 2002
-
- Graham GG, Graham RI, Day RO. Comparative analgesia, cardiovascular and renal effects of celecoxib, rofecoxib and acetaminophen (paracetamol). Current Pharmaceutical Design 2002;8(12):1063‐75. - PubMed
Graham 2003
-
- Graham GG, Scott KF. Mechanisms of action of paracetamol and related analgesics. Inflammopharmacology 2003;11(4):401‐13. - PubMed
Graham 2013
-
- Graham GG, Davies MJ, Day RO, Mohamudally A, Scott KF. The modern pharmacology of paracetamol: therapeutic actions, mechanism of action, metabolism, toxicity and recent pharmacological findings. Inflammopharmacology 2013;21(3):201‐32. - PubMed
Guyatt 2008
-
- Guyatt GH, Cook DJ, Jaeschke R, Pauker SG, Schünemann HJ. Grades of recommendation for antithrombotic agents: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th Edition). Chest 2008;133(6):123S‐31S. - PubMed
Guyatt 2011
-
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction – GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64(4):383‐94. - PubMed
Gøtzsche 2007
-
- Gøtzsche PC, Hróbjartsson A, Maric K, Tendal B. Data extraction errors in meta‐analyses that use standardized mean differences. JAMA 2007;298(4):430‐7. - PubMed
Higgins 2011
-
- Higgins JP, Altman DG, Sterne JA. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Hinz 2008
-
- Hinz B, Cheremina O, Brune K. Acetaminophen (paracetamol) is a selective cyclooxygenase‐2 inhibitor in man. FASEB Journal 2008;22(2):383‐90. - PubMed
Hochberg 2012
-
- Hochberg MC, Altman RD, April KT, Benkhalti M, Guyatt G, McGowan J, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care and Research 2012;64:465‐74. - PubMed
Hoy 2014
-
- Hoy D, March L, Brooks P, Blyth F, Woolf A, Bain C, et al. The global burden of low back pain: estimates from the Global Burden of Disease 2010 study. Annals of the Rheumatic Diseases 2014;73:968‐74. - PubMed
Jordan 2003
-
- Jordan KM, Arden NK, Doherty M, Bannwarth B, Bijlsma JW, Dieppe P, et al. EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Annals of the Rheumatic Diseases 2003;62(12):1145‐55. - PMC - PubMed
Juhl 2012
Jüni 2006
-
- Jüni P, Reichenbach S, Dieppe P. Osteoarthritis: rational approach to treating the individual. Best Practice and Research 2006;20(4):720‐40. - PubMed
Jüni 2015
Machado 2015
Pham 2004
-
- Pham T, Heijde D, Altman RD, Anderson JJ, Bellamy N, Hochberg M, et al. OMERACT‐OARSI initiative: Osteoarthritis Research Society International set of responder criteria for osteoarthritis clinical trials revisited. Osteoarthritis and Cartilage 2004;12(5):389‐99. - PubMed
Puljak 2017
Regnaux 2015
-
- Regnaux JP, Lefevre‐Colau MM, Trinquart L, Nguyen C, Boutron I, Brosseau L, et al. High‐intensity versus low‐intensity physical activity or exercise in people with hip or knee osteoarthritis. Cochrane Database of Systematic Reviews 2015, Issue 10. [DOI: 10.1002/14651858.CD010203.pub2] - DOI - PMC - PubMed
Reichenbach 2007
-
- Reichenbach S, Sterchi R, Scherer M, Trelle S, Burgi E, Burgi U, et al. Meta‐analysis: chondroitin for osteoarthritis of the knee or hip. Annals of Internal Medicine 2007;146(8):580‐90. - PubMed
Review Manager 2012 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.2. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2012.
Roberts 2016
Schein 2008
-
- Schein JR, Kosinski MR, Janagap‐Benson C, Gajria K, Lin P, Freedman JD. Functionality and health‐status benefits associated with reduction of osteoarthritis pain. Current Medical Research and Opinion 2008;24:1255‐65. - PubMed
Smink 2011
-
- Smink AJ, Ende CH, Vliet Vlieland TP, Swierstra BA, Kortland JH, Bijlsma JW, et al. "Beating osteoARThritis": development of a stepped care strategy to optimize utilization and timing of non‐surgical treatment modalities for patients with hip or knee osteoarthritis. Clinical Rheumatology 2011;30(12):1623‐9. - PubMed
Towheed 2006
Uthman 2013
Vos 2012
Wandel 2010
Woolf 2003
Zhang 2004
Zhang 2005
-
- Zhang W, Doherty M, Arden N, Bannwarth B, Bijlsma J, Gunther KP, et al. EULAR evidence based recommendations for the management of hip osteoarthritis: report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Annals of the Rheumatic Diseases 2005;64:669‐81. - PMC - PubMed
Zhang 2007
-
- Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, et al. OARSI recommendations for the management of hip and knee osteoarthritis, part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis and Cartilage 2007;15(19):981‐1000. - PubMed
Zhang 2010
-
- Zhang WG, Nuki RW, Moskowitz S, Abramson RD, Altman NK, Arden S, et al. OARSI recommendations for the management of hip and knee osteoarthritis: part III: changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis and Cartilage 2010;18(4):476‐99. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

