Modified quadruple therapy versus bismuth-containing quadruple therapy in first-line treatment of Helicobacter pylori infection in Korea; rationale and design of an open-label, multicenter, randomized controlled trial
- PMID: 30431605
- PMCID: PMC6257664
- DOI: 10.1097/MD.0000000000013245
Modified quadruple therapy versus bismuth-containing quadruple therapy in first-line treatment of Helicobacter pylori infection in Korea; rationale and design of an open-label, multicenter, randomized controlled trial
Abstract
Background: Clarithromycin-containing triple regimen for eradication of Helicobacter pylori is no longer acceptable in Korea due to high clarithromycin resistance. Concomitant therapy or bismuth-containing quadruple therapy is recommended as an alternative regimen. A recent study in Korea has shown that modified quadruple therapy has comparable efficacy and safety to concomitant therapy as a first-line regimen. However, there has been no comparative study of modified quadruple therapy with bismuth-containing quadruple therapy. The aim of this study is to compare the efficacy and safety of modified quadruple therapy with those of bismuth-containing quadruple therapy as a first-line regimen and to present the phenotypic and genotypic antibiotic resistance profile of H pylori.
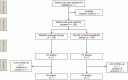
Methods: This study is an open-label, multicenter, randomized controlled trial. We are recruiting subjects endoscopically diagnosed with H pylori infection from 2 hospitals in Korea. Subjects will be randomly allocated either to modified quadruple therapy (proton-pump inhibitor bid, amoxicillin 1 g bid, metronidazole 500 mg tid, bismuth subcitrate 300 mg qid daily) or bismuth-containing quadruple therapy (proton-pump inhibitor bid, tetracycline 500 mg qid, metronidazole 500 mg tid, bismuth subcitrate 300 mg qid daily) for 14 days. The rate of eradication success and adverse events will be checked at least 4 weeks after the treatment. Antibiotic resistance will be established using both a bacterial culture with agar dilutions and DNA sequencing of the clarithromycin resistance point mutations in the 23S rRNA gene of H pylori.
Conclusion: The results of this study will provide solid evidence for determining the optimal treatment regimen for first-line H pylori eradication in Korea.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures
Similar articles
-
Amoxicillin or tetracycline in bismuth-containing quadruple therapy as first-line treatment for Helicobacter pylori infection.Gut Microbes. 2020 Sep 2;11(5):1314-1323. doi: 10.1080/19490976.2020.1754118. Epub 2020 May 2. Gut Microbes. 2020. PMID: 32362221 Free PMC article. Clinical Trial.
-
Efficacy comparison of 7- and 14-day P-CAB based bismuth-containing quadruple regimen with PPI based bismuth-containing quadruple regimen for Helicobacter pylori infection: rationale and design of an open-label, multicenter, randomized controlled trial.BMC Gastroenterol. 2023 Dec 21;23(1):453. doi: 10.1186/s12876-023-03100-y. BMC Gastroenterol. 2023. PMID: 38129806 Free PMC article.
-
Concomitant, bismuth quadruple, and 14-day triple therapy in the first-line treatment of Helicobacter pylori: a multicentre, open-label, randomised trial.Lancet. 2016 Nov 12;388(10058):2355-2365. doi: 10.1016/S0140-6736(16)31409-X. Epub 2016 Oct 18. Lancet. 2016. PMID: 27769562 Clinical Trial.
-
The Toronto Consensus for the Treatment of Helicobacter pylori Infection in Adults.Gastroenterology. 2016 Jul;151(1):51-69.e14. doi: 10.1053/j.gastro.2016.04.006. Epub 2016 Apr 19. Gastroenterology. 2016. PMID: 27102658 Review.
-
Optimum duration of regimens for Helicobacter pylori eradication.Cochrane Database Syst Rev. 2013 Dec 11;2013(12):CD008337. doi: 10.1002/14651858.CD008337.pub2. Cochrane Database Syst Rev. 2013. PMID: 24338763 Free PMC article. Review.
Cited by
-
Amoxicillin or tetracycline in bismuth-containing quadruple therapy as first-line treatment for Helicobacter pylori infection.Gut Microbes. 2020 Sep 2;11(5):1314-1323. doi: 10.1080/19490976.2020.1754118. Epub 2020 May 2. Gut Microbes. 2020. PMID: 32362221 Free PMC article. Clinical Trial.
References
-
- Kwak HW, Choi IJ, Cho SJ, et al. Characteristics of gastric cancer according to Helicobacter pylori infection status. J Gastroenterol Hepatol 2014;29:1671–7. - PubMed
-
- Correa P. A human model of gastric carcinogenesis. Cancer Res 1988;48:3554–60. - PubMed
-
- Choi IJ, Kook MC, Kim YI, et al. Helicobacter pylori therapy for the prevention of metachronous gastric cancer. N Engl J Med 2018;378:1085–95. - PubMed
-
- Lee YC, Chiang TH, Chou CK, et al. Association between Helicobacter pylori eradication and gastric cancer incidence: a systematic review and meta-analysis. Gastroenterology 2016;150:1113–24. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical