Trafficking of ciliary membrane proteins by the intraflagellar transport/BBSome machinery
- PMID: 30287585
- PMCID: PMC6737936
- DOI: 10.1042/EBC20180030
Trafficking of ciliary membrane proteins by the intraflagellar transport/BBSome machinery
Abstract
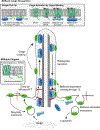
Bardet-Biedl syndrome (BBS) is a rare inherited disease caused by defects in the BBSome, an octameric complex of BBS proteins. The BBSome is conserved in most organisms with cilia, which are microtubule (MT)-based cell organelles that protrude from the cell surface and function in motility and sensing. Cilia assembly, maintenance, and function require intraflagellar transport (IFT), a bidirectional motility of multi-megadalton IFT trains propelled by molecular motors along the ciliary MTs. IFT has been shown to transport structural proteins, including tubulin, into growing cilia. The BBSome is an adapter for the transport of ciliary membrane proteins and cycles through cilia via IFT. While both the loss and the abnormal accumulation of ciliary membrane proteins have been observed in bbs mutants, recent data converge on a model where the BBSome mainly functions as a cargo adapter for the removal of certain transmembrane and peripheral membrane proteins from cilia. Here, we review recent data on the ultrastructure of the BBSome and how the BBSome recognizes its cargoes and mediates their removal from cilia.
Keywords: BBSome; G-protein-coupled receptors; cilia; hedgehog; intraflagellar transport; membrane proteins.
© 2018 The Author(s). Published by Portland Press Limited on behalf of the Biochemical Society.
Conflict of interest statement
Figures
Similar articles
-
The Bardet-Biedl syndrome protein complex is an adapter expanding the cargo range of intraflagellar transport trains for ciliary export.Proc Natl Acad Sci U S A. 2018 Jan 30;115(5):E934-E943. doi: 10.1073/pnas.1713226115. Epub 2018 Jan 16. Proc Natl Acad Sci U S A. 2018. PMID: 29339469 Free PMC article.
-
Ciliary protein trafficking mediated by IFT and BBSome complexes with the aid of kinesin-2 and dynein-2 motors.J Biochem. 2018 Mar 1;163(3):155-164. doi: 10.1093/jb/mvx087. J Biochem. 2018. PMID: 29272450 Review.
-
Whole-Organism Developmental Expression Profiling Identifies RAB-28 as a Novel Ciliary GTPase Associated with the BBSome and Intraflagellar Transport.PLoS Genet. 2016 Dec 8;12(12):e1006469. doi: 10.1371/journal.pgen.1006469. eCollection 2016 Dec. PLoS Genet. 2016. PMID: 27930654 Free PMC article.
-
Cargo adapters expand the transport range of intraflagellar transport.J Cell Sci. 2022 Dec 15;135(24):jcs260408. doi: 10.1242/jcs.260408. Epub 2022 Dec 19. J Cell Sci. 2022. PMID: 36533425 Free PMC article. Review.
-
Unraveling the intricate cargo-BBSome coupling mechanism at the ciliary tip.Proc Natl Acad Sci U S A. 2023 Mar 28;120(13):e2218819120. doi: 10.1073/pnas.2218819120. Epub 2023 Mar 21. Proc Natl Acad Sci U S A. 2023. PMID: 36943875 Free PMC article.
Cited by
-
Ccrk-Mak/Ick signaling is a ciliary transport regulator essential for retinal photoreceptor survival.Life Sci Alliance. 2024 Sep 18;7(11):e202402880. doi: 10.26508/lsa.202402880. Print 2024 Nov. Life Sci Alliance. 2024. PMID: 39293864 Free PMC article.
-
The exocyst complex and intracellular vesicles mediate soluble protein trafficking to the primary cilium.Commun Biol. 2024 Feb 21;7(1):213. doi: 10.1038/s42003-024-05817-2. Commun Biol. 2024. PMID: 38378792 Free PMC article.
-
Ciliary Proteins: Filling the Gaps. Recent Advances in Deciphering the Protein Composition of Motile Ciliary Complexes.Cells. 2019 Jul 17;8(7):730. doi: 10.3390/cells8070730. Cells. 2019. PMID: 31319499 Free PMC article. Review.
-
Rab35 controls cilium length, function and membrane composition.EMBO Rep. 2019 Oct 4;20(10):e47625. doi: 10.15252/embr.201847625. Epub 2019 Aug 21. EMBO Rep. 2019. PMID: 31432619 Free PMC article.
-
BBS7-SHH Signaling Activity Regulates Primary Cilia for Periodontal Homeostasis.Front Cell Dev Biol. 2021 Dec 7;9:796274. doi: 10.3389/fcell.2021.796274. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34957122 Free PMC article.
References
-
- Benzinou M, Walley A, Lobbens S, Charles MA, Jouret B, Fumeron F, et al. Bardet-Biedl syndrome gene variants are associated with both childhood and adult common obesity in French Caucasians. Diabetes 2006;55(10):2876–82. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

