Elderly dendritic cells respond to LPS/IFN-γ and CD40L stimulation despite incomplete maturation
- PMID: 29652910
- PMCID: PMC5898732
- DOI: 10.1371/journal.pone.0195313
Elderly dendritic cells respond to LPS/IFN-γ and CD40L stimulation despite incomplete maturation
Abstract
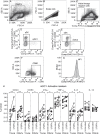
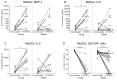
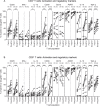
There is evidence that dendritic cells (DCs) undergo age-related changes that modulate their function with their key role being priming antigen-specific effector T cells. This occurs once DCs develop into antigen-presenting cells in response to stimuli/danger signals. However, the effects of aging on DC responses to bacterial lipopolysaccharide (LPS), the pro-inflammatory cytokine interferon (IFN)-γ and CD40 ligand (CD40L) have not yet been systematically evaluated. We examined responses of blood myeloid (m)DC1s, mDC2s, plasmacytoid (p)DCs, and monocyte-derived DCs (MoDCs) from young (21-40 years) and elderly (60-84 years) healthy human volunteers to LPS/IFN-γ or CD40L stimulation. All elderly DC subsets demonstrated comparable up-regulation of co-stimulatory molecules (CD40, CD80 and/or CD86), intracellular pro-inflammatory cytokine levels (IFN-γ, tumour necrosis factor (TNF)-α, IL-6 and/or IL-12), and/or secreted cytokine levels (IFN-α, IFN-γ, TNF-α, and IL-12) to their younger counterparts. Furthermore, elderly-derived LPS/IFN-γ or CD40L-activated MoDCs induced similar or increased levels of CD8+ and CD4+ T cell proliferation, and similar T cell functional phenotypes, to their younger counterparts. However, elderly LPS/IFN-γ-activated MoDCs were unreliable in their ability to up-regulate chemokine (IL-8 and monocyte chemoattractant protein (MCP)-1) and IL-6 secretion, implying an inability to dependably induce an inflammatory response. A key age-related difference was that, unlike young-derived MoDCs that completely lost their ability to process antigen, elderly-derived MoDCs maintained their antigen processing ability after LPS/IFN-γ maturation, measured using the DQ-ovalbumin assay; this response implies incomplete maturation that may enable elderly DCs to continuously present antigen. These differences may impact on the efficacy of anti-pathogen and anti-tumour immune responses in the elderly.
Conflict of interest statement
Figures



Similar articles
-
The influence of CD40 ligation and interferon-gamma on functional properties of human monocyte-derived dendritic cells activated with polyinosinic-polycytidylic acid.Vojnosanit Pregl. 2011 Apr;68(4):301-8. doi: 10.2298/vsp1104301d. Vojnosanit Pregl. 2011. PMID: 21627015
-
Sequential delivery of maturation stimuli increases human dendritic cell IL-12 production and enhances tumor antigen-specific immunogenicity.J Surg Res. 2004 Jan;116(1):24-31. doi: 10.1016/j.jss.2003.09.003. J Surg Res. 2004. PMID: 14732346
-
IL-21 enhances SOCS gene expression and inhibits LPS-induced cytokine production in human monocyte-derived dendritic cells.J Leukoc Biol. 2006 Jun;79(6):1279-85. doi: 10.1189/jlb.0905503. Epub 2006 Mar 21. J Leukoc Biol. 2006. PMID: 16551679
-
The role of CD40 expression in dendritic cells in cancer biology; a systematic review.Curr Cancer Drug Targets. 2014;14(7):610-20. doi: 10.2174/1568009614666140828103253. Curr Cancer Drug Targets. 2014. PMID: 25163469 Review.
-
Altered cytokine production in the elderly.Mech Ageing Dev. 1998 May 15;102(2-3):199-209. doi: 10.1016/s0047-6374(97)00153-x. Mech Ageing Dev. 1998. PMID: 9720652 Review.
Cited by
-
Bifidobacterium bifidum and Bacteroides fragilis Induced Differential Immune Regulation of Enteric Glial Cells Subjected to Exogenous Inflammatory Stimulation.Inflammation. 2022 Dec;45(6):2388-2405. doi: 10.1007/s10753-022-01700-6. Epub 2022 Jul 1. Inflammation. 2022. PMID: 35776290
-
Kidney transplantation in the elderly.Korean J Intern Med. 2024 Nov;39(6):875-881. doi: 10.3904/kjim.2024.089. Epub 2024 Oct 24. Korean J Intern Med. 2024. PMID: 39444336 Free PMC article. Review.
-
The Impact of Sphingosine Kinases on Inflammation-Induced Cytokine Release and Vascular Endothelial Barrier Integrity.Int J Mol Sci. 2022 Oct 25;23(21):12848. doi: 10.3390/ijms232112848. Int J Mol Sci. 2022. PMID: 36361636 Free PMC article.
-
The Nature of Immune Responses to Influenza Vaccination in High-Risk Populations.Viruses. 2021 Jun 9;13(6):1109. doi: 10.3390/v13061109. Viruses. 2021. PMID: 34207924 Free PMC article. Review.
-
Inhibition of DAI refrains dendritic cells from maturation and prolongs murine islet and skin allograft survival.Front Immunol. 2023 May 1;14:1182851. doi: 10.3389/fimmu.2023.1182851. eCollection 2023. Front Immunol. 2023. PMID: 37197662 Free PMC article.
References
-
- Wordsworth D, Dunn-Walters D. The ageing immune system and its clinical implications. Rev Clin Gerontol. 2011;21(02):110–24. doi: 10.1017/S0959259810000407 - DOI
-
- Lee N, Shin MS, Kang I. T-cell biology in aging, with a focus on lung disease. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences. 2012;67A(3):254–63. doi: 10.1093/gerona/glr237 - DOI - PMC - PubMed
-
- Haynes L, Maue AC. Effects of aging on T cell function. Curr Opin Immunol. 2009;21(4):414–7. doi: 10.1016/j.coi.2009.05.009 - DOI - PMC - PubMed
-
- Gupta S. Role of dendritic cells in innate and adaptive immune response in human aging. Exp Gerontol. 2014;54:47–52. doi: 10.1016/j.exger.2013.12.009 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

