Comparison of Helicobacter pylori Eradication Rates of 2-Week Levofloxacin-Containing Triple Therapy, Levofloxacin-Containing Bismuth Quadruple Therapy, and Standard Bismuth Quadruple Therapy as a First-Line Regimen
- PMID: 29131124
- PMCID: PMC5848476
- DOI: 10.1159/000484930
Comparison of Helicobacter pylori Eradication Rates of 2-Week Levofloxacin-Containing Triple Therapy, Levofloxacin-Containing Bismuth Quadruple Therapy, and Standard Bismuth Quadruple Therapy as a First-Line Regimen
Abstract
Objective: The aim of this study was to compare the efficacy and safety of 2-week levofloxacin-containing triple therapy, levofloxacin-containing bismuth quadruple therapy, and standard bismuth-containing quadruple therapy as a first-line regimen for the eradication of Helicobacter pylori.
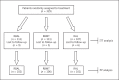
Methods: A total of 329 patients with H. pylori infection were randomly divided into 3 groups to receive one of the following regimens: (a) levofloxacin-containing bismuth quadruple therapy, RBAL (rabeprazole 20 mg, b.i.d., bismuth subsalicylate 562 mg, b.i.d., amoxicillin 1 g, b.i.d, levofloxacin 500 mg, once daily), (b) standard bismuth quadruple therapy, RBMT (rabeprazole 20 mg, b.i.d, subsalicylate 562 mg, b.i.d., metronidazole 500 mg, t.i.d, tetracycline 500 mg, q.i.d), or (c) levofloxacin-containing triple therapy, RAL (rabeprazole 20 mg, b.i.d., amoxicillin 1 g, b.i.d, levofloxacin 500 mg, once daily). The primary outcome was the eradication rate in the intention-to-treat (ITT) and per protocol (PP) analysis.
Results: The eradication rates of the above 3 groups using ITT analysis were RBAL 83.8%, RBMT 88.3%, and RAL 74.8% compared with 91.2, 92.5, and 79.2%, respectively, using PP analysis. The eradication rate using RBMT was significantly higher than that of RAL (p = 0.029 in ITT analysis and p = 0.017 in PP analysis). Several side effects occurred in 156 patients (54.1%) in the RBAL group, 215 (52.3%) in the RBMT group, and 56 (26.2%) in the RAL group (p > 0.05, RBAL vs. RBMT; p < 0.001, RBMT vs. RAL; p < 0.001, RBAL vs. RAL).
Conclusion: All bismuth-containing quadruple therapies had acceptable eradication rates, but levofloxacin-containing triple therapy was not as good as quadruple therapies. Hence, quadruple therapies should be considered the preferred first-line therapy for H. pylori infections.
Keywords:
© 2017 The Author(s) Published by S. Karger AG, Basel.
Figures
Similar articles
-
Comparison of 10-day levofloxacin bismuth-based quadruple therapy and levofloxacin-based triple therapy for Helicobacter pylori.J Dig Dis. 2017 Sep;18(9):537-542. doi: 10.1111/1751-2980.12498. J Dig Dis. 2017. PMID: 28644575 Clinical Trial.
-
Comparison of bismuth-containing quadruple and concomitant therapies as a first-line treatment option for Helicobacter pylori.Turk J Gastroenterol. 2012 Feb;23(1):8-13. doi: 10.4318/tjg.2012.0392. Turk J Gastroenterol. 2012. PMID: 22505373 Clinical Trial.
-
Amoxicillin or tetracycline in bismuth-containing quadruple therapy as first-line treatment for Helicobacter pylori infection.Gut Microbes. 2020 Sep 2;11(5):1314-1323. doi: 10.1080/19490976.2020.1754118. Epub 2020 May 2. Gut Microbes. 2020. PMID: 32362221 Free PMC article. Clinical Trial.
-
Second-line rescue treatment of Helicobacter pylori infection: Where are we now?World J Gastroenterol. 2018 Oct 28;24(40):4548-4553. doi: 10.3748/wjg.v24.i40.4548. World J Gastroenterol. 2018. PMID: 30386104 Free PMC article. Review.
-
A new look at anti-Helicobacter pylori therapy.World J Gastroenterol. 2011 Sep 21;17(35):3971-5. doi: 10.3748/wjg.v17.i35.3971. World J Gastroenterol. 2011. PMID: 22046084 Free PMC article. Review.
Cited by
-
Levofloxacin+Tetracycline Quadruple Regimen for Eradication of Helicobacter pylori: A Multicenter Multinational Randomized Controlled Trial.Middle East J Dig Dis. 2023 Jan;15(1):12-18. doi: 10.34172/mejdd.2023.314. Epub 2023 Jan 30. Middle East J Dig Dis. 2023. PMID: 37547163 Free PMC article.
-
2022 Chinese national clinical practice guideline on Helicobacter pylori eradication treatment.Chin Med J (Engl). 2022 Dec 20;135(24):2899-2910. doi: 10.1097/CM9.0000000000002546. Chin Med J (Engl). 2022. PMID: 36579940 Free PMC article.
-
Can the treatment duration be shortened in bismuth-containing therapies for Helicobacter pylori eradication?Turk J Gastroenterol. 2019 Aug;30(8):667-672. doi: 10.5152/tjg.2019.18793. Turk J Gastroenterol. 2019. PMID: 31258136 Free PMC article. Clinical Trial.
-
Reevaluation of the Efficacy of First Line Regimen for Helicobacter pylori.Clin Exp Gastroenterol. 2020 Jan 22;13:25-33. doi: 10.2147/CEG.S239343. eCollection 2020. Clin Exp Gastroenterol. 2020. PMID: 32158248 Free PMC article.
-
Rabeprazole-amoxicillin dual therapy as first-line treatment for H pylori eradication in special patients: A retrospective, real-life study.Helicobacter. 2020 Oct;25(5):e12717. doi: 10.1111/hel.12717. Epub 2020 Jun 16. Helicobacter. 2020. PMID: 32548932 Free PMC article.
References
-
- Kamboj AK, Cotter TG, Oxentenko AS. Helicobacter pylori: the past, present, and future in management. Mayo Clin Proc. 2017;92:599–604. - PubMed
-
- Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347:1175–1186. - PubMed
-
- Malfertheiner P, Bazzolli F. Helicobacter pylori eradication with a capsule containing bismuth subcitrate potassium, metronidazole, and tetracycline given with omeprazole versus clarithromycin-based triple therapy: a randomized, open-label, non-inferiority, phase 3 trial. Lancet. 2011;377:905–913. - PubMed
-
- Fischbach L, Evans EL. Meta-analysis: the effect of antibiotic resistance status on the efficacy of triple and quadruple first-line therapies for Helicobacter pylori. Aliment Pharmacol Ther. 2007;26:343e57. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

