BolA Is Required for the Accurate Regulation of c-di-GMP, a Central Player in Biofilm Formation
- PMID: 28928205
- PMCID: PMC5605933
- DOI: 10.1128/mBio.00443-17
BolA Is Required for the Accurate Regulation of c-di-GMP, a Central Player in Biofilm Formation
Abstract
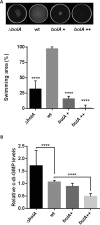
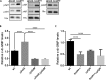
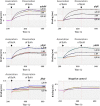
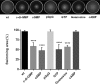
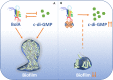
The bacterial second messenger cyclic dimeric GMP (c-di-GMP) is a nearly ubiquitous intracellular signaling molecule involved in the transition from the motile to the sessile/biofilm state in bacteria. C-di-GMP regulates various cellular processes, including biofilm formation, motility, and virulence. BolA is a transcription factor that promotes survival in different stresses and is also involved in biofilm formation. Both BolA and c-di-GMP participate in the regulation of motility mechanisms leading to similar phenotypes. Here, we establish the importance of the balance between these two factors for accurate regulation of the transition between the planktonic and sessile lifestyles. This balance is achieved by negative-feedback regulation of BolA and c-di-GMP. BolA not only contributes directly to the motility of bacteria but also regulates the expression of diguanylate cyclases and phosphodiesterases. This expression modulation influences the synthesis and degradation of c-di-GMP, while this signaling metabolite has a negative influence in bolA mRNA transcription. Finally, we present evidence of the dominant role of BolA in biofilm, showing that, even in the presence of elevated c-di-GMP levels, biofilm formation is reduced in the absence of BolA. C-di-GMP is one of the most important bacterial second messengers involved in several cellular processes, including virulence, cell cycle regulation, biofilm formation, and flagellar synthesis. In this study, we unravelled a direct connection between the bolA morphogene and the c-di-GMP signaling molecule. We show the important cross-talk that occurs between these two molecular regulators during the transition between the motile/planktonic and adhesive/sessile lifestyles in Escherichia coli This work provides important clues that can be helpful in the development of new strategies, and the results can be applied to other organisms with relevance for human health.IMPORTANCE Bacterial cells have evolved several mechanisms to cope with environmental stresses. BolA-like proteins are widely conserved from prokaryotes to eukaryotes, and in Escherichia coli, in addition to its pleiotropic effects, this protein plays a determinant role in bacterial motility and biofilm formation regulation. Similarly, the bacterial second messenger c-di-GMP is a molecule with high importance in coordinating the switch between planktonic and sessile life in bacteria. Here we have unravelled the importance of accurate regulation of cross-talk between BolA and c-di-GMP for a proper response in the regulation of these bacterial lifestyles. This finding underlines the complexity of bacterial cell regulation, revealing the existence of one additional tool for fine-tuning such important cellular molecular mechanisms. The relationship between BolA and c-di-GMP gives new perspectives regarding biofilm formation and opens the possibility to extend our studies to other organisms with relevance for human health.
Keywords: BolA; biofilm; c-di-GMP; diguanylate cyclase; motility; phosphodiesterase.
Copyright © 2017 Moreira et al.
Figures
Similar articles
-
Expression and Genetic Activation of Cyclic Di-GMP-Specific Phosphodiesterases in Escherichia coli.J Bacteriol. 2015 Nov 9;198(3):448-62. doi: 10.1128/JB.00604-15. Print 2016 Feb 1. J Bacteriol. 2015. PMID: 26553851 Free PMC article.
-
Bordetella bronchiseptica Diguanylate Cyclase BdcA Regulates Motility and Is Important for the Establishment of Respiratory Infection in Mice.J Bacteriol. 2019 Aug 8;201(17):e00011-19. doi: 10.1128/JB.00011-19. Print 2019 Sep 1. J Bacteriol. 2019. PMID: 31209073 Free PMC article.
-
BolA is a transcriptional switch that turns off motility and turns on biofilm development.mBio. 2015 Feb 17;6(1):e02352-14. doi: 10.1128/mBio.02352-14. mBio. 2015. PMID: 25691594 Free PMC article.
-
The bacterial second messenger c-di-GMP: probing interactions with protein and RNA binding partners using cyclic dinucleotide analogs.Org Biomol Chem. 2012 Dec 14;10(46):9113-29. doi: 10.1039/c2ob26724a. Epub 2012 Oct 29. Org Biomol Chem. 2012. PMID: 23108253 Free PMC article. Review.
-
Targeting c-di-GMP Signaling, Biofilm Formation, and Bacterial Motility with Small Molecules.Methods Mol Biol. 2017;1657:419-430. doi: 10.1007/978-1-4939-7240-1_31. Methods Mol Biol. 2017. PMID: 28889311 Review.
Cited by
-
Preventing Biofilm Formation and Development on Ear, Nose and Throat Medical Devices.Biomedicines. 2021 Aug 16;9(8):1025. doi: 10.3390/biomedicines9081025. Biomedicines. 2021. PMID: 34440229 Free PMC article. Review.
-
BolA-like protein (IbaG) promotes biofilm formation and pathogenicity of Vibrio parahaemolyticus.Front Microbiol. 2024 Jul 31;15:1436770. doi: 10.3389/fmicb.2024.1436770. eCollection 2024. Front Microbiol. 2024. PMID: 39144210 Free PMC article.
-
Phosphorylated OmpR Is Required for Type 3 Fimbriae Expression in Klebsiella pneumoniae Under Hypertonic Conditions.Front Microbiol. 2018 Oct 12;9:2405. doi: 10.3389/fmicb.2018.02405. eCollection 2018. Front Microbiol. 2018. PMID: 30369914 Free PMC article.
-
Identification of the BolA Protein Reveals a Novel Virulence Factor in K. pneumoniae That Contributes to Survival in Host.Microbiol Spectr. 2022 Oct 26;10(5):e0037822. doi: 10.1128/spectrum.00378-22. Epub 2022 Sep 19. Microbiol Spectr. 2022. PMID: 36121239 Free PMC article.
-
The Catabolite Repressor/Activator Cra Is a Bridge Connecting Carbon Metabolism and Host Colonization in the Plant Drought Resistance-Promoting Bacterium Pantoea alhagi LTYR-11Z.Appl Environ Microbiol. 2018 Jun 18;84(13):e00054-18. doi: 10.1128/AEM.00054-18. Print 2018 Jul 1. Appl Environ Microbiol. 2018. PMID: 29703735 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
