RAC1 Missense Mutations in Developmental Disorders with Diverse Phenotypes
- PMID: 28886345
- PMCID: PMC5591022
- DOI: 10.1016/j.ajhg.2017.08.007
RAC1 Missense Mutations in Developmental Disorders with Diverse Phenotypes
Abstract
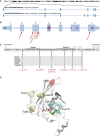
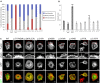
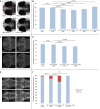
RAC1 is a widely studied Rho GTPase, a class of molecules that modulate numerous cellular functions essential for normal development. RAC1 is highly conserved across species and is under strict mutational constraint. We report seven individuals with distinct de novo missense RAC1 mutations and varying degrees of developmental delay, brain malformations, and additional phenotypes. Four individuals, each harboring one of c.53G>A (p.Cys18Tyr), c.116A>G (p.Asn39Ser), c.218C>T (p.Pro73Leu), and c.470G>A (p.Cys157Tyr) variants, were microcephalic, with head circumferences between -2.5 to -5 SD. In contrast, two individuals with c.151G>A (p.Val51Met) and c.151G>C (p.Val51Leu) alleles were macrocephalic with head circumferences of +4.16 and +4.5 SD. One individual harboring a c.190T>G (p.Tyr64Asp) allele had head circumference in the normal range. Collectively, we observed an extraordinary spread of ∼10 SD of head circumferences orchestrated by distinct mutations in the same gene. In silico modeling, mouse fibroblasts spreading assays, and in vivo overexpression assays using zebrafish as a surrogate model demonstrated that the p.Cys18Tyr and p.Asn39Ser RAC1 variants function as dominant-negative alleles and result in microcephaly, reduced neuronal proliferation, and cerebellar abnormalities in vivo. Conversely, the p.Tyr64Asp substitution is constitutively active. The remaining mutations are probably weakly dominant negative or their effects are context dependent. These findings highlight the importance of RAC1 in neuronal development. Along with TRIO and HACE1, a sub-category of rare developmental disorders is emerging with RAC1 as the central player. We show that ultra-rare disorders caused by private, non-recurrent missense mutations that result in varying phenotypes are challenging to dissect, but can be delineated through focused international collaboration.
Keywords: HACE1; RAC1; Rho GTPase; TRIO; cerebellar abnormalities; developmental disorders; intellectual disability; macrocephaly; microcephaly; neuronal proliferation.
Copyright © 2017 American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Opposite Modulation of RAC1 by Mutations in TRIO Is Associated with Distinct, Domain-Specific Neurodevelopmental Disorders.Am J Hum Genet. 2020 Mar 5;106(3):338-355. doi: 10.1016/j.ajhg.2020.01.018. Epub 2020 Feb 27. Am J Hum Genet. 2020. PMID: 32109419 Free PMC article.
-
Bi-allelic Variants in DYNC1I2 Cause Syndromic Microcephaly with Intellectual Disability, Cerebral Malformations, and Dysmorphic Facial Features.Am J Hum Genet. 2019 Jun 6;104(6):1073-1087. doi: 10.1016/j.ajhg.2019.04.002. Epub 2019 May 9. Am J Hum Genet. 2019. PMID: 31079899 Free PMC article.
-
Germline mutations affecting the histone H4 core cause a developmental syndrome by altering DNA damage response and cell cycle control.Nat Genet. 2017 Nov;49(11):1642-1646. doi: 10.1038/ng.3956. Epub 2017 Sep 18. Nat Genet. 2017. PMID: 28920961
-
Novel NGLY1 gene variants in Chinese children with global developmental delay, microcephaly, hypotonia, hypertransaminasemia, alacrimia, and feeding difficulty.J Hum Genet. 2020 Apr;65(4):387-396. doi: 10.1038/s10038-019-0719-9. Epub 2020 Jan 21. J Hum Genet. 2020. PMID: 31965062 Review.
-
Extending the phenotypes associated with TRIO gene variants in a cohort of 25 patients and review of the literature.Am J Med Genet A. 2023 Jul;191(7):1722-1740. doi: 10.1002/ajmg.a.63194. Epub 2023 Mar 29. Am J Med Genet A. 2023. PMID: 36987741 Review.
Cited by
-
Active components and molecular mechanisms of Sagacious Confucius' Pillow Elixir to treat cognitive impairment based on systems pharmacology.Aging (Albany NY). 2023 Jul 30;15(14):7278-7307. doi: 10.18632/aging.204912. Epub 2023 Jul 30. Aging (Albany NY). 2023. PMID: 37517091 Free PMC article.
-
Rho GTPase Regulators and Effectors in Autism Spectrum Disorders: Animal Models and Insights for Therapeutics.Cells. 2020 Mar 31;9(4):835. doi: 10.3390/cells9040835. Cells. 2020. PMID: 32244264 Free PMC article. Review.
-
De Novo Variants in SPOP Cause Two Clinically Distinct Neurodevelopmental Disorders.Am J Hum Genet. 2020 Mar 5;106(3):405-411. doi: 10.1016/j.ajhg.2020.02.001. Epub 2020 Feb 27. Am J Hum Genet. 2020. PMID: 32109420 Free PMC article.
-
Case report: A gain-of-function of hamartin may lead to a distinct "inverse TSC1-hamartin" phenotype characterized by reduced cell growth.Front Pediatr. 2023 Mar 30;11:1101026. doi: 10.3389/fped.2023.1101026. eCollection 2023. Front Pediatr. 2023. PMID: 37063680 Free PMC article.
-
Presynaptic Rac1 controls synaptic strength through the regulation of synaptic vesicle priming.Elife. 2022 Oct 10;11:e81505. doi: 10.7554/eLife.81505. Elife. 2022. PMID: 36214784 Free PMC article.
References
-
- Sheridan E., Wright J., Small N., Corry P.C., Oddie S., Whibley C., Petherick E.S., Malik T., Pawson N., McKinney P.A., Parslow R.C. Risk factors for congenital anomaly in a multiethnic birth cohort: an analysis of the Born in Bradford study. Lancet. 2013;382:1350–1359. - PubMed
-
- Ropers H.H. Genetics of early onset cognitive impairment. Annu. Rev. Genomics Hum. Genet. 2010;11:161–187. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

