Mitigation of a nitrate reducing Pseudomonas aeruginosa biofilm and anaerobic biocorrosion using ciprofloxacin enhanced by D-tyrosine
- PMID: 28761161
- PMCID: PMC5537228
- DOI: 10.1038/s41598-017-07312-7
Mitigation of a nitrate reducing Pseudomonas aeruginosa biofilm and anaerobic biocorrosion using ciprofloxacin enhanced by D-tyrosine
Abstract
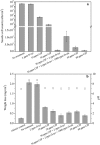
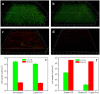
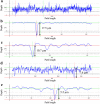
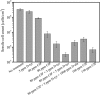
Pseudomonas aeruginosa (PA) is a ubiquitous microbe. It can form recalcitrant biofilms in clinical and industrial settings. PA biofilms cause infections in patients. They also cause biocorrosion of medical implants. In this work, D-tyrosine (D-tyr) was investigated as an antimicrobial enhancer for ciprofloxacin (CIP) against a wild-type PA biofilm (strain PAO1) on C1018 carbon steel in a strictly anaerobic condition. Seven-day biofilm prevention test results demonstrated that 2 ppm (w/w) D-tyr enhanced 30 ppm CIP by achieving extra 2-log sessile cell reduction compared with the 30 ppm CIP alone treatment. The cocktail of 30 ppm CIP + 2 ppm D-tyr achieved similar efficacy as the 80 ppm CIP alone treatment in the biofilm prevention test. Results also indicated that the enhanced antimicrobial treatment reduced weight loss and pitting corrosion. In the 3-hour biofilm removal test, the cocktail of 80 ppm CIP + 5 ppm D-tyr achieved extra 1.5-log reduction in sessile cell count compared with the 80 ppm CIP alone treatment. The cocktail of 80 ppm CIP + 5 ppm D-tyr achieved better efficacy than the 150 ppm CIP alone treatment in the biofilm removal test.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



Similar articles
-
Mitigation of galvanized steel biocorrosion by Pseudomonas aeruginosa biofilm using a biocide enhanced by trehalase.Bioelectrochemistry. 2023 Dec;154:108508. doi: 10.1016/j.bioelechem.2023.108508. Epub 2023 Jul 10. Bioelectrochemistry. 2023. PMID: 37451042
-
Efficacy of Ciprofloxacin and Its Copper Complex against Pseudomonas aeruginosa Biofilms.AAPS PharmSciTech. 2019 May 29;20(5):205. doi: 10.1208/s12249-019-1417-9. AAPS PharmSciTech. 2019. PMID: 31144198
-
Lack of the Major Multifunctional Catalase KatA in Pseudomonas aeruginosa Accelerates Evolution of Antibiotic Resistance in Ciprofloxacin-Treated Biofilms.Antimicrob Agents Chemother. 2019 Sep 23;63(10):e00766-19. doi: 10.1128/AAC.00766-19. Print 2019 Oct. Antimicrob Agents Chemother. 2019. PMID: 31307984 Free PMC article.
-
Extracellular Electron Transfer by Pseudomonas aeruginosa in Biocorrosion: A Review.ACS Biomater Sci Eng. 2022 Mar 14;8(3):1049-1059. doi: 10.1021/acsbiomaterials.1c01645. Epub 2022 Feb 24. ACS Biomater Sci Eng. 2022. PMID: 35199512 Review.
-
Biosurfactants: Eco-Friendly and Innovative Biocides against Biocorrosion.Int J Mol Sci. 2020 Mar 20;21(6):2152. doi: 10.3390/ijms21062152. Int J Mol Sci. 2020. PMID: 32245097 Free PMC article. Review.
Cited by
-
Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention.Heliyon. 2018 Dec 28;4(12):e01067. doi: 10.1016/j.heliyon.2018.e01067. eCollection 2018 Dec. Heliyon. 2018. PMID: 30619958 Free PMC article. Review.
-
Beyond Risk: Bacterial Biofilms and Their Regulating Approaches.Front Microbiol. 2020 May 21;11:928. doi: 10.3389/fmicb.2020.00928. eCollection 2020. Front Microbiol. 2020. PMID: 32508772 Free PMC article. Review.
-
Anaerobic Corrosion of 304 Stainless Steel Caused by the Pseudomonas aeruginosa Biofilm.Front Microbiol. 2017 Nov 27;8:2335. doi: 10.3389/fmicb.2017.02335. eCollection 2017. Front Microbiol. 2017. PMID: 29230206 Free PMC article.
-
Promising Application of D-Amino Acids toward Clinical Therapy.Int J Mol Sci. 2022 Sep 16;23(18):10794. doi: 10.3390/ijms231810794. Int J Mol Sci. 2022. PMID: 36142706 Free PMC article. Review.
-
Enhanced Biocide Treatments with D-amino Acid Mixtures against a Biofilm Consortium from a Water Cooling Tower.Front Microbiol. 2017 Aug 16;8:1538. doi: 10.3389/fmicb.2017.01538. eCollection 2017. Front Microbiol. 2017. PMID: 28861053 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

