Movement disorder in GNAO1 encephalopathy associated with gain-of-function mutations
- PMID: 28747448
- PMCID: PMC5580866
- DOI: 10.1212/WNL.0000000000004262
Movement disorder in GNAO1 encephalopathy associated with gain-of-function mutations
Abstract
Objective: To define molecular mechanisms underlying the clinical spectrum of epilepsy and movement disorder in individuals with de novo mutations in the GNAO1 gene.
Methods: We identified all GNAO1 mutations reported in individuals with epilepsy (early infantile epileptiform encephalopathy 17) or movement disorders through April 2016; 15 de novo mutant alleles from 25 individuals were introduced into the Gαo subunit by site-directed mutagenesis in a mammalian expression plasmid. We assessed protein expression and function in vitro in HEK-293T cells by Western blot and determined functional Gαo-dependent cyclic adenosine monophosphate (cAMP) inhibition with a coexpressed α2A adrenergic receptor.
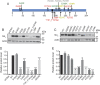
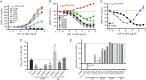
Results: Of the 15 clinical GNAO1 mutations studied, 9 show reduced expression and loss of function (LOF; <90% maximal inhibition). Six other mutations show variable levels of expression but exhibit normal or even gain-of-function (GOF) behavior, as demonstrated by significantly lower EC50 values for α2A adrenergic receptor-mediated inhibition of cAMP. The GNAO1 LOF mutations are associated with epileptic encephalopathy while GOF mutants (such as G42R, G203R, and E246K) or normally functioning mutants (R209) were found in patients with movement disorders with or without seizures.
Conclusions: Both LOF and GOF mutations in Gαo (encoded by GNAO1) are associated with neurologic pathophysiology. There appears to be a strong predictive correlation between the in vitro biochemical phenotype and the clinical pattern of epilepsy vs movement disorder.
© 2017 American Academy of Neurology.
Figures
Comment in
-
Epileptic encephalopathy, movement disorder, and the yin and yang of GNAO1 function.Neurology. 2017 Aug 22;89(8):754-755. doi: 10.1212/WNL.0000000000004277. Epub 2017 Jul 26. Neurology. 2017. PMID: 28747451 No abstract available.
Similar articles
-
Mouse models of GNAO1-associated movement disorder: Allele- and sex-specific differences in phenotypes.PLoS One. 2019 Jan 25;14(1):e0211066. doi: 10.1371/journal.pone.0211066. eCollection 2019. PLoS One. 2019. Retraction in: PLoS One. 2021 Oct 14;16(10):e0258912. doi: 10.1371/journal.pone.0258912. PMID: 30682176 Free PMC article. Retracted.
-
A mechanistic review on GNAO1-associated movement disorder.Neurobiol Dis. 2018 Aug;116:131-141. doi: 10.1016/j.nbd.2018.05.005. Epub 2018 May 24. Neurobiol Dis. 2018. PMID: 29758257 Review.
-
Mouse models characterize GNAO1 encephalopathy as a neurodevelopmental disorder leading to motor anomalies: from a severe G203R to a milder C215Y mutation.Acta Neuropathol Commun. 2022 Jan 28;10(1):9. doi: 10.1186/s40478-022-01312-z. Acta Neuropathol Commun. 2022. PMID: 35090564 Free PMC article.
-
Clinical and Molecular Profiling in GNAO1 Permits Phenotype-Genotype Correlation.Mov Disord. 2024 Sep;39(9):1578-1591. doi: 10.1002/mds.29881. Epub 2024 Jun 16. Mov Disord. 2024. PMID: 38881224
-
Phenomenology and clinical course of movement disorder in GNAO1 variants: Results from an analytical review.Parkinsonism Relat Disord. 2019 Apr;61:19-25. doi: 10.1016/j.parkreldis.2018.11.019. Epub 2018 Nov 16. Parkinsonism Relat Disord. 2019. PMID: 30642806 Review.
Cited by
-
Postsynaptic movement disorders: clinical phenotypes, genotypes, and disease mechanisms.J Inherit Metab Dis. 2018 Nov;41(6):1077-1091. doi: 10.1007/s10545-018-0205-0. Epub 2018 Jun 13. J Inherit Metab Dis. 2018. PMID: 29948482 Free PMC article. Review.
-
Restoration of the GTPase activity and cellular interactions of Gαo mutants by Zn2+ in GNAO1 encephalopathy models.Sci Adv. 2022 Oct 7;8(40):eabn9350. doi: 10.1126/sciadv.abn9350. Epub 2022 Oct 7. Sci Adv. 2022. PMID: 36206333 Free PMC article.
-
The Schwann cell-specific G-protein Gαo (Gnao1) is a cell-intrinsic controller contributing to the regulation of myelination in peripheral nerve system.Acta Neuropathol Commun. 2024 Feb 8;12(1):24. doi: 10.1186/s40478-024-01720-3. Acta Neuropathol Commun. 2024. PMID: 38331815 Free PMC article.
-
Humanization of Drosophila Gαo to Model GNAO1 Paediatric Encephalopathies.Biomedicines. 2020 Oct 6;8(10):395. doi: 10.3390/biomedicines8100395. Biomedicines. 2020. PMID: 33036271 Free PMC article.
-
Molecular Dynamic Simulations to Determine Individualized Therapy: Tetrabenazine for the GNAO1 Encephalopathy E246K Variant.Mol Diagn Ther. 2024 May;28(3):329-337. doi: 10.1007/s40291-024-00706-0. Epub 2024 Apr 6. Mol Diagn Ther. 2024. PMID: 38581611
References
-
- Kobau R, Ly ZM, Helmers S, Thurman D. Epilepsy in adults and access to care: United States. MMWR Morb Mortal Wkly Rep 2012;61:909–931. - PubMed
-
- Capovilla G, Wolf P, Beccaria F, Avanzini G. The history of the concept of epileptic encephalopathy. Epilepsia 2013;54(suppl 8):2–5. - PubMed
-
- McTague A, Howell KB, Cross JH, Kurian MA, Scheffer IE. The genetic landscape of the epileptic encephalopathies of infancy and childhood. Lancet Neurol 2016;15:304–316. - PubMed
-
- Sherr EH. The ARX story (epilepsy, mental retardation, autism, and cerebral malformations): one gene leads to many phenotypes. Curr Opin Pediatr 2003;15:567–571. - PubMed
-
- Berkovic SF, Heron SE, Giordano L, et al. . Benign familial neonatal-infantile seizures: characterization of a new sodium channelopathy. Ann Neurol 2004;55:550–557. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
