Randomized Trial of C5a Receptor Inhibitor Avacopan in ANCA-Associated Vasculitis
- PMID: 28400446
- PMCID: PMC5576933
- DOI: 10.1681/ASN.2016111179
Randomized Trial of C5a Receptor Inhibitor Avacopan in ANCA-Associated Vasculitis
Abstract
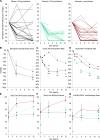
Alternative C activation is involved in the pathogenesis of ANCA-associated vasculitis. However, glucocorticoids used as treatment contribute to the morbidity and mortality of vasculitis. We determined whether avacopan (CCX168), an orally administered, selective C5a receptor inhibitor, could replace oral glucocorticoids without compromising efficacy. In this randomized, placebo-controlled trial, adults with newly diagnosed or relapsing vasculitis received placebo plus prednisone starting at 60 mg daily (control group), avacopan (30 mg, twice daily) plus reduced-dose prednisone (20 mg daily), or avacopan (30 mg, twice daily) without prednisone. All patients received cyclophosphamide or rituximab. The primary efficacy measure was the proportion of patients achieving a ≥50% reduction in Birmingham Vasculitis Activity Score by week 12 and no worsening in any body system. We enrolled 67 patients, 23 in the control and 22 in each of the avacopan groups. Clinical response at week 12 was achieved in 14 of 20 (70.0%) control patients, 19 of 22 (86.4%) patients in the avacopan plus reduced-dose prednisone group (difference from control 16.4%; two-sided 90% confidence limit, -4.3% to 37.1%; P=0.002 for noninferiority), and 17 of 21 (81.0%) patients in the avacopan without prednisone group (difference from control 11.0%; two-sided 90% confidence limit, -11.0% to 32.9%; P=0.01 for noninferiority). Adverse events occurred in 21 of 23 (91%) control patients, 19 of 22 (86%) patients in the avacopan plus reduced-dose prednisone group, and 21 of 22 (96%) patients in the avacopan without prednisone group. In conclusion, C5a receptor inhibition with avacopan was effective in replacing high-dose glucocorticoids in treating vasculitis.
Keywords: ANCA; ANCA-associated vasculitis; avacopan; complement; complement 5a; complement 5a receptor.
Copyright © 2017 by the American Society of Nephrology.
Figures
Similar articles
-
Avacopan for the Treatment of ANCA-Associated Vasculitis.N Engl J Med. 2021 Feb 18;384(7):599-609. doi: 10.1056/NEJMoa2023386. N Engl J Med. 2021. PMID: 33596356 Clinical Trial.
-
Avacopan in the treatment of ANCA-associated vasculitis.Expert Opin Investig Drugs. 2018 May;27(5):491-496. doi: 10.1080/13543784.2018.1472234. Epub 2018 May 8. Expert Opin Investig Drugs. 2018. PMID: 29718732 Review.
-
Avacopan, a selective C5a receptor antagonist, for anti-neutrophil cytoplasmic antibody-associated vasculitis.Mod Rheumatol. 2022 Apr 18;32(3):475-483. doi: 10.1093/mr/roab104. Mod Rheumatol. 2022. PMID: 34984461
-
Avacopan: First Approval.Drugs. 2022 Jan;82(1):79-85. doi: 10.1007/s40265-021-01643-6. Drugs. 2022. PMID: 34826105 Review.
-
Glucocorticoid Toxicity Index scores by domain in patients with antineutrophil cytoplasmic antibody-associated vasculitis treated with avacopan versus standard prednisone taper: post-hoc analysis of data from the ADVOCATE trial.Lancet Rheumatol. 2023 Mar;5(3):e130-e138. doi: 10.1016/S2665-9913(23)00030-9. Epub 2023 Feb 20. Lancet Rheumatol. 2023. PMID: 38251609 Clinical Trial.
Cited by
-
Therapeutic Targeting of the Complement System: From Rare Diseases to Pandemics.Pharmacol Rev. 2021 Apr;73(2):792-827. doi: 10.1124/pharmrev.120.000072. Pharmacol Rev. 2021. PMID: 33687995 Free PMC article.
-
Recent advances in understanding of the pathogenesis of ANCA-associated vasculitis.F1000Res. 2018 Jul 19;7:F1000 Faculty Rev-1113. doi: 10.12688/f1000research.14626.1. eCollection 2018. F1000Res. 2018. PMID: 30079228 Free PMC article. Review.
-
Animal models for anti-neutrophil cytoplasmic antibody-associated vasculitis: Are current models good enough?Animal Model Exp Med. 2023 Oct;6(5):452-463. doi: 10.1002/ame2.12345. Epub 2023 Aug 23. Animal Model Exp Med. 2023. PMID: 37614099 Free PMC article. Review.
-
The complement system as a potential therapeutic target in rheumatic disease.Nat Rev Rheumatol. 2017 Sep;13(9):538-547. doi: 10.1038/nrrheum.2017.125. Epub 2017 Aug 10. Nat Rev Rheumatol. 2017. PMID: 28794515 Review.
-
Eosinophilic Granulomatosis With Polyangiitis: Dissecting the Pathophysiology.Front Med (Lausanne). 2021 Feb 24;8:627776. doi: 10.3389/fmed.2021.627776. eCollection 2021. Front Med (Lausanne). 2021. PMID: 33718405 Free PMC article. Review.
References
-
- Luqmani R, Suppiah R, Edwards CJ, Phillip R, Maskell J, Culliford D, Jayne D, Morishita K, Arden N: Mortality in Wegener’s granulomatosis: A bimodal pattern. Rheumatology (Oxford) 50: 697–702, 2011 - PubMed
-
- Little MA, Nightingale P, Verburgh CA, Hauser T, De Groot K, Savage C, Jayne D, Harper L; European Vasculitis Study (EUVAS) Group : Early mortality in systemic vasculitis: Relative contribution of adverse events and active vasculitis. Ann Rheum Dis 69: 1036–1043, 2010 - PubMed
-
- Jayne D: Evidence-based treatment of systemic vasculitis. Rheumatology (Oxford) 39: 585–595, 2000 - PubMed
-
- Koldingsnes W, Nossent H: Predictors of survival and organ damage in Wegener’s granulomatosis. Rheumatology (Oxford) 41: 572–581, 2002 - PubMed
-
- Corral-Gudino L, Borao-Cengotita-Bengoa M, Del Pino-Montes J, Lerma-Márquez JL: Overall survival, renal survival and relapse in patients with microscopic polyangiitis: A systematic review of current evidence. Rheumatology (Oxford) 50: 1414–1423, 2011 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

