Efficacy and Safety of Anti-Trop-2 Antibody Drug Conjugate Sacituzumab Govitecan (IMMU-132) in Heavily Pretreated Patients With Metastatic Triple-Negative Breast Cancer
- PMID: 28291390
- PMCID: PMC5559902
- DOI: 10.1200/JCO.2016.70.8297
Efficacy and Safety of Anti-Trop-2 Antibody Drug Conjugate Sacituzumab Govitecan (IMMU-132) in Heavily Pretreated Patients With Metastatic Triple-Negative Breast Cancer
Abstract
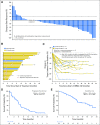
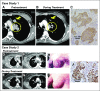
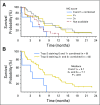
Purpose Trop-2, expressed in most triple-negative breast cancers (TNBCs), may be a potential target for antibody-drug conjugates. Sacituzumab govitecan, an antibody-drug conjugate, targets Trop-2 for the selective delivery of SN-38, the active metabolite of irinotecan. Patients and Methods We evaluated sacituzumab govitecan in a single-arm, multicenter trial in patients with relapsed/refractory metastatic TNBC who received a 10 mg/kg starting dose on days 1 and 8 of 21-day repeated cycles. The primary end points were safety and objective response rate; secondary end points were progression-free survival and overall survival. Results In 69 patients who received a median of five prior therapies (range, one to 12) since diagnosis, the confirmed objective response rate was 30% (partial response, n = 19; complete response, n = 2), the median response duration was 8.9 (95% CI, 6.1 to 11.3) months, and the clinical benefit rate (complete response + partial response + stable disease ≥ 6 months) was 46%. These responses occurred early, with a median onset of 1.9 months. Median progression-free survival was 6.0 (95% CI, 5.0 to 7.3) months, and median overall survival was 16.6 (95% CI, 11.1 to 20.6) months. Grade ≥ 3 adverse events included neutropenia (39%), leukopenia (16%), anemia (14%), and diarrhea (13%); the incidence of febrile neutropenia was 7%. The majority of archival tumor specimens (88%) were moderately to strongly positive for Trop-2 by immunohistochemistry. No neutralizing antibodies to the ADC or antibody were detected, despite repeated cycles developed. Conclusion Sacituzumab govitecan was well tolerated and induced early and durable responses in heavily pretreated patients with metastatic TNBC. As a therapeutic target and predictive biomarker, Trop-2 warrants further research.
Figures

Similar articles
-
Therapy of Advanced Non-Small-Cell Lung Cancer With an SN-38-Anti-Trop-2 Drug Conjugate, Sacituzumab Govitecan.J Clin Oncol. 2017 Aug 20;35(24):2790-2797. doi: 10.1200/JCO.2016.72.1894. Epub 2017 May 26. J Clin Oncol. 2017. PMID: 28548889 Clinical Trial.
-
Sacituzumab Govitecan-hziy in Refractory Metastatic Triple-Negative Breast Cancer.N Engl J Med. 2019 Feb 21;380(8):741-751. doi: 10.1056/NEJMoa1814213. N Engl J Med. 2019. PMID: 30786188 Clinical Trial.
-
Sacituzumab govitecan (IMMU-132), an anti-Trop-2-SN-38 antibody-drug conjugate for the treatment of diverse epithelial cancers: Safety and pharmacokinetics.Cancer. 2017 Oct 1;123(19):3843-3854. doi: 10.1002/cncr.30789. Epub 2017 May 30. Cancer. 2017. PMID: 28558150 Clinical Trial.
-
Sacituzumab govitecan: breakthrough targeted therapy for triple-negative breast cancer.Expert Rev Anticancer Ther. 2019 Aug;19(8):673-679. doi: 10.1080/14737140.2019.1654378. Epub 2019 Aug 19. Expert Rev Anticancer Ther. 2019. PMID: 31398063 Review.
-
Sacituzumab for the treatment of triple-negative breast cancer: the poster child of future therapy?Expert Opin Investig Drugs. 2019 Feb;28(2):107-112. doi: 10.1080/13543784.2019.1555239. Epub 2018 Dec 17. Expert Opin Investig Drugs. 2019. PMID: 30507322 Review.
Cited by
-
The Role of TROP2 in BCC and Cutaneous SCC: A Clinical and Immunohistochemical Study.Clin Cosmet Investig Dermatol. 2021 Jun 1;14:591-600. doi: 10.2147/CCID.S299862. eCollection 2021. Clin Cosmet Investig Dermatol. 2021. PMID: 34103959 Free PMC article.
-
Triple-negative breast cancer: current perspective on the evolving therapeutic landscape.Int J Womens Health. 2019 Jul 31;11:431-437. doi: 10.2147/IJWH.S178349. eCollection 2019. Int J Womens Health. 2019. PMID: 31447592 Free PMC article.
-
Theranostics for Triple-Negative Breast Cancer.Diagnostics (Basel). 2023 Jan 11;13(2):272. doi: 10.3390/diagnostics13020272. Diagnostics (Basel). 2023. PMID: 36673082 Free PMC article. Review.
-
Antibody-Drug Conjugates for the Treatment of Breast Cancer.Cancers (Basel). 2021 Jun 9;13(12):2898. doi: 10.3390/cancers13122898. Cancers (Basel). 2021. PMID: 34207890 Free PMC article. Review.
-
Trop-2 protein as a therapeutic target: A focused review on Trop-2-based antibody-drug conjugates and their predictive biomarkers.Bosn J Basic Med Sci. 2022 Feb 1;22(1):14-21. doi: 10.17305/bjbms.2021.6100. Bosn J Basic Med Sci. 2022. PMID: 34181512 Free PMC article. Review.
References
-
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. - PubMed
-
- Perou CM, Sørlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. - PubMed
-
- Carey LA, Perou CM, Livasy CA, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–2502. - PubMed
-
- Hurvitz S, Mead M. Triple-negative breast cancer: Advancements in characterization and treatment approach. Curr Opin Obstet Gynecol. 2016;28:59–69. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

