Bacopa monnieri extract increases rat coronary flow and protects against myocardial ischemia/reperfusion injury
- PMID: 28219356
- PMCID: PMC5319078
- DOI: 10.1186/s12906-017-1637-z
Bacopa monnieri extract increases rat coronary flow and protects against myocardial ischemia/reperfusion injury
Abstract
Background: This study explored Bacopa monnieri, a medicinal Ayurvedic herb, as a cardioprotectant against ischemia/reperfusion injury using cardiac function and coronary flow as end-points.
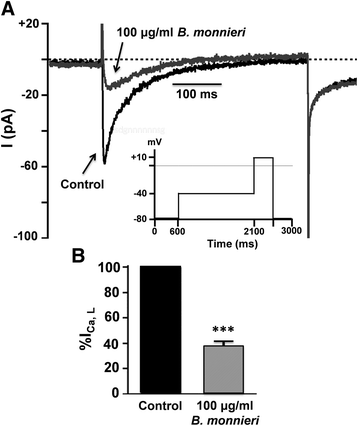
Methods: In normal isolated rat hearts, coronary flow, left ventricular developed pressure, heart rate, and functional recovery were measured using the Langendorff preparation. Hearts were perfused with either (i) Krebs-Henseleit (normal) solution, (control), or with 30, 100 μg/ml B. monnieri ethanolic extract (30 min), or (ii) with normal solution or extract for 10 min preceding no-perfusion ischemia (30 min) followed by reperfusion (30 min) with normal solution. Infarct volumes were measured by triphenyltetrazolium staining. L-type Ca2+-currents (ICa, L) were measured by whole-cell patching in HL-1 cells, a mouse atrial cardiomyocyte cell line. Cytotoxicity of B. monnieri was assessed in rat isolated ventricular myocytes by trypan blue exclusion.
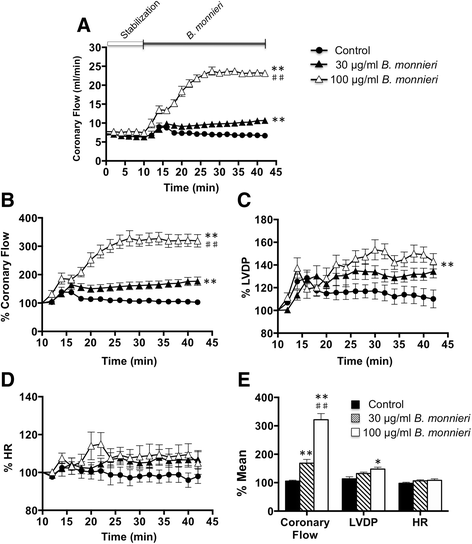
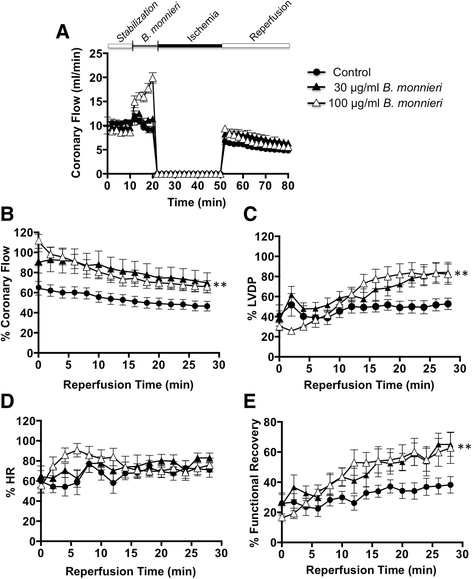
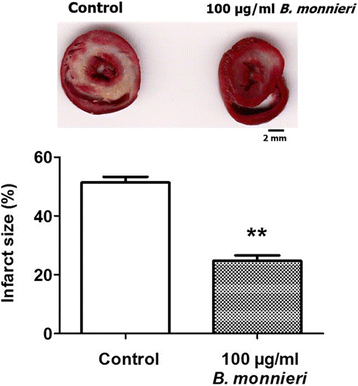
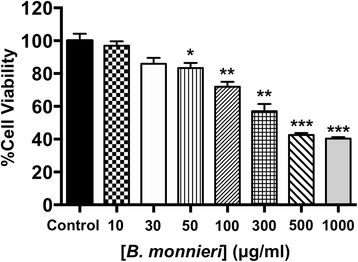
Results: In normally perfused hearts, B. monnieri increased coronary flow by 63 ± 13% (30 μg/ml) and 216 ± 21% (100 μg/ml), compared to control (5 ± 3%) (n = 8-10, p < 0.001). B. monnieri treatment preceding ischemia/reperfusion improved left ventricular developed pressure by 84 ± 10% (30 μg/ml), 82 ± 10% (100 μg/ml) and 52 ± 6% (control) compared to pre- ischemia/reperfusion. Similarly, functional recovery showed a sustained increase. Moreover, B. monnieri (100 μg/ml) reduced the percentage of infarct size from 51 ± 2% (control) to 25 ± 2% (n = 6-8, p < 0.0001). B. monnieri (100 μg/ml) reduced ICa, L by 63 ± 4% in HL-1 cells. Ventricular myocyte survival decreased at higher concentrations (50-1000 μg/ml) B. monnieri.
Conclusions: B. monnieri improves myocardial function following ischemia/reperfusion injury through recovery of coronary blood flow, contractile force and decrease in infarct size. Thus this may lead to a novel cardioprotectant strategy.
Keywords: Bacopa monnieri; Brahmi; Cardiac function; Coronary blood flow; Heart; Ischemia/reperfusion; Myocardial infarction.
Figures
Similar articles
-
Salvia miltiorrhiza treatment during early reperfusion reduced postischemic myocardial injury in the rat.Can J Physiol Pharmacol. 2007 Oct;85(10):1012-9. doi: 10.1139/y07-092. Can J Physiol Pharmacol. 2007. PMID: 18066102
-
Bacopa monniera protects rat heart against ischaemia-reperfusion injury: role of key apoptotic regulatory proteins and enzymes.J Pharm Pharmacol. 2010 Sep;62(9):1175-84. doi: 10.1111/j.2042-7158.2010.01155.x. J Pharm Pharmacol. 2010. PMID: 20796197
-
Phenylephrine induces early and late cardioprotection through mitochondrial permeability transition pore in the isolated rat heart.J Surg Res. 2010 Nov;164(1):e37-42. doi: 10.1016/j.jss.2010.04.060. Epub 2010 May 26. J Surg Res. 2010. PMID: 20850771
-
The protective role of curcumin in myocardial ischemia-reperfusion injury.J Cell Physiol. 2018 Jan;234(1):214-222. doi: 10.1002/jcp.26848. Epub 2018 Jul 3. J Cell Physiol. 2018. PMID: 29968913 Review.
-
Does reperfusion injury exist in humans?J Am Coll Cardiol. 1993 Feb;21(2):537-45. doi: 10.1016/0735-1097(93)90700-b. J Am Coll Cardiol. 1993. PMID: 8426022 Review.
Cited by
-
Protective action of the ginsenoside Rh3 in a rat myocardial ischemia-reperfusion injury model by inhibition of apoptosis induced via p38 mitogen-activated protein kinase/caspase-3 signaling.J Int Med Res. 2020 Dec;48(12):300060520969090. doi: 10.1177/0300060520969090. J Int Med Res. 2020. Retraction in: J Int Med Res. 2024 Jan;52(1):3000605241228165. doi: 10.1177/03000605241228165. PMID: 33284724 Free PMC article. Retracted.
-
Advances in research on the protective mechanisms of traditional Chinese medicine (TCM) in myocardial ischaemia-reperfusion injury.Pharm Biol. 2022 Dec;60(1):931-948. doi: 10.1080/13880209.2022.2063342. Pharm Biol. 2022. PMID: 35587352 Free PMC article.
-
Vasodilatory Effects and Mechanisms of Action of Bacopa monnieri Active Compounds on Rat Mesenteric Arteries.Molecules. 2019 Jun 15;24(12):2243. doi: 10.3390/molecules24122243. Molecules. 2019. PMID: 31208086 Free PMC article.
-
Protective Effects of Galium verum L. Extract against Cardiac Ischemia/Reperfusion Injury in Spontaneously Hypertensive Rats.Oxid Med Cell Longev. 2019 Feb 4;2019:4235405. doi: 10.1155/2019/4235405. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 30863479 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

