Moringa oleifera as an Anti-Cancer Agent against Breast and Colorectal Cancer Cell Lines
- PMID: 26288313
- PMCID: PMC4545797
- DOI: 10.1371/journal.pone.0135814
Moringa oleifera as an Anti-Cancer Agent against Breast and Colorectal Cancer Cell Lines
Abstract
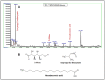
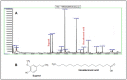
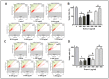
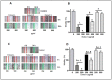
In this study we investigated the anti-cancer effect of Moringa oleifera leaves, bark and seed extracts. When tested against MDA-MB-231 and HCT-8 cancer cell lines, the extracts of leaves and bark showed remarkable anti-cancer properties while surprisingly, seed extracts exhibited hardly any such properties. Cell survival was significantly low in both cells lines when treated with leaves and bark extracts. Furthermore, a striking reduction (about 70-90%) in colony formation as well as cell motility was observed upon treatment with leaves and bark. Additionally, apoptosis assay performed on these treated breast and colorectal cancer lines showed a remarkable increase in the number of apoptotic cells; with a 7 fold increase in MD-MB-231 to an increase of several fold in colorectal cancer cell lines. However, no significant apoptotic cells were detected upon seeds extract treatment. Moreover, the cell cycle distribution showed a G2/M enrichment (about 2-3 fold) indicating that these extracts effectively arrest the cell progression at the G2/M phase. The GC-MS analyses of these extracts revealed numerous known anti-cancer compounds, namely eugenol, isopropyl isothiocynate, D-allose, and hexadeconoic acid ethyl ester, all of which possess long chain hydrocarbons, sugar moiety and an aromatic ring. This suggests that the anti-cancer properties of Moringa oleifera could be attributed to the bioactive compounds present in the extracts from this plant. This is a novel study because no report has yet been cited on the effectiveness of Moringa extracts obtained in the locally grown environment as an anti-cancer agent against breast and colorectal cancers. Our study is the first of its kind to evaluate the anti-malignant properties of Moringa not only in leaves but also in bark. These findings suggest that both the leaf and bark extracts of Moringa collected from the Saudi Arabian region possess anti-cancer activity that can be used to develop new drugs for treatment of breast and colorectal cancers.
Conflict of interest statement
Figures


Similar articles
-
Moringa peregrina Leaves Extracts Induce Apoptosis and Cell Cycle Arrest of Hepatocellular Carcinoma.Biomed Res Int. 2019 Jan 1;2019:2698570. doi: 10.1155/2019/2698570. eCollection 2019. Biomed Res Int. 2019. PMID: 30713850 Free PMC article.
-
Cytotoxic activity of extracts and crude saponins from Zanthoxylum armatum DC. against human breast (MCF-7, MDA-MB-468) and colorectal (Caco-2) cancer cell lines.BMC Complement Altern Med. 2017 Jul 17;17(1):368. doi: 10.1186/s12906-017-1882-1. BMC Complement Altern Med. 2017. PMID: 28716103 Free PMC article.
-
Significant Decreased Expressions of CaN, VEGF, SLC39A6 and SFRP1 in MDA-MB-231 Xenograft Breast Tumor Mice Treated with Moringa oleifera Leaves and Seed Residue (MOLSr) Extracts.Nutrients. 2020 Sep 30;12(10):2993. doi: 10.3390/nu12102993. Nutrients. 2020. PMID: 33007803 Free PMC article.
-
Moringa oleifera Seeds and Oil: Characteristics and Uses for Human Health.Int J Mol Sci. 2016 Dec 20;17(12):2141. doi: 10.3390/ijms17122141. Int J Mol Sci. 2016. PMID: 27999405 Free PMC article. Review.
-
Review of the Safety and Efficacy of Moringa oleifera.Phytother Res. 2015 Jun;29(6):796-804. doi: 10.1002/ptr.5325. Epub 2015 Mar 24. Phytother Res. 2015. PMID: 25808883 Free PMC article. Review.
Cited by
-
The use of African medicinal plants in cancer management.Front Pharmacol. 2023 Feb 14;14:1122388. doi: 10.3389/fphar.2023.1122388. eCollection 2023. Front Pharmacol. 2023. PMID: 36865913 Free PMC article. Review.
-
Scorpion Venom Causes Upregulation of p53 and Downregulation of Bcl-xL and BID Protein Expression by Modulating Signaling Proteins Erk1/2 and STAT3, and DNA Damage in Breast and Colorectal Cancer Cell Lines.Integr Cancer Ther. 2018 Jun;17(2):271-281. doi: 10.1177/1534735417704949. Epub 2017 Apr 25. Integr Cancer Ther. 2018. PMID: 28438053 Free PMC article.
-
Snake venom causes apoptosis by increasing the reactive oxygen species in colorectal and breast cancer cell lines.Onco Targets Ther. 2016 Oct 20;9:6485-6498. doi: 10.2147/OTT.S115055. eCollection 2016. Onco Targets Ther. 2016. PMID: 27799796 Free PMC article.
-
Evaluation of wound healing properties of bioactive aqueous fraction from Moringa oleifera Lam on experimentally induced diabetic animal model.Drug Des Devel Ther. 2016 May 24;10:1715-30. doi: 10.2147/DDDT.S96968. eCollection 2016. Drug Des Devel Ther. 2016. PMID: 27307703 Free PMC article.
-
Gene expression and spatiotemporal localization of antifungal chitin-binding proteins during Moringa oleifera seed development and germination.Planta. 2019 May;249(5):1503-1519. doi: 10.1007/s00425-019-03103-8. Epub 2019 Jan 31. Planta. 2019. PMID: 30706136
References
-
- Olson ME. Combining data from DNA sequences and morphology for a phylogeny of Moringaceae (Brassicales). Systematic Botany. 2002;27(1):55–73.
-
- Paulo Michel Pinheiro Ferreira, Éverton José Ferreira de Araújo, Jurandy do Nascimento Silva, Rivelilson Mendes de Freitas, Nagilla Daniela de Jesus Costa, Samara Ferreira de Carvalho Oliveira, et al. Safety and Efficacy of Moringa oleifera Lamarck (1785)—Therapeutic and Toxicological Properties. In: Gowder DSJT, editor. Pharmacology and Therapeutics,: InTech; 2014.
-
- Patel S, Thakur A, Chandy A, Manigauha A. Moringa Oleifera: A Review of There Medicinal and Economical Importance to the Health and Nation. Drug invention today. 2010;2(7).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

