Clinical and biochemical characterization of four patients with mutations in ECHS1
- PMID: 26081110
- PMCID: PMC4474341
- DOI: 10.1186/s13023-015-0290-1
Clinical and biochemical characterization of four patients with mutations in ECHS1
Abstract
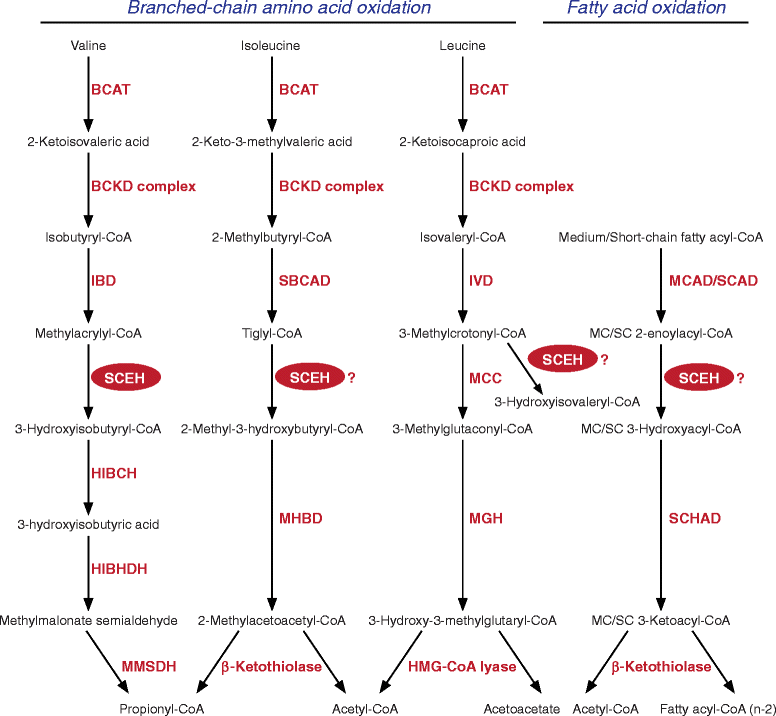
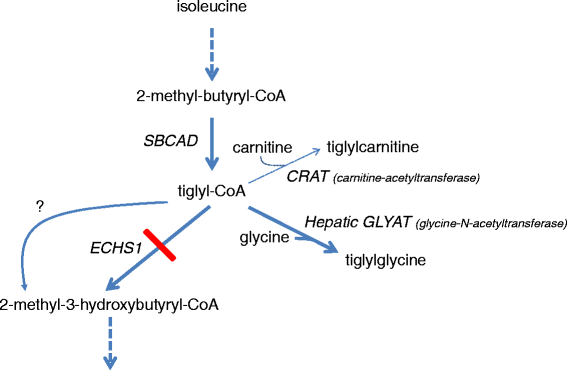
Background: Short-chain enoyl-CoA hydratase (SCEH, encoded by ECHS1) catalyzes hydration of 2-trans-enoyl-CoAs to 3(S)-hydroxy-acyl-CoAs. SCEH has a broad substrate specificity and is believed to play an important role in mitochondrial fatty acid oxidation and in the metabolism of branched-chain amino acids. Recently, the first patients with SCEH deficiency have been reported revealing only a defect in valine catabolism. We investigated the role of SCEH in fatty acid and branched-chain amino acid metabolism in four newly identified patients. In addition, because of the Leigh-like presentation, we studied enzymes involved in bioenergetics.
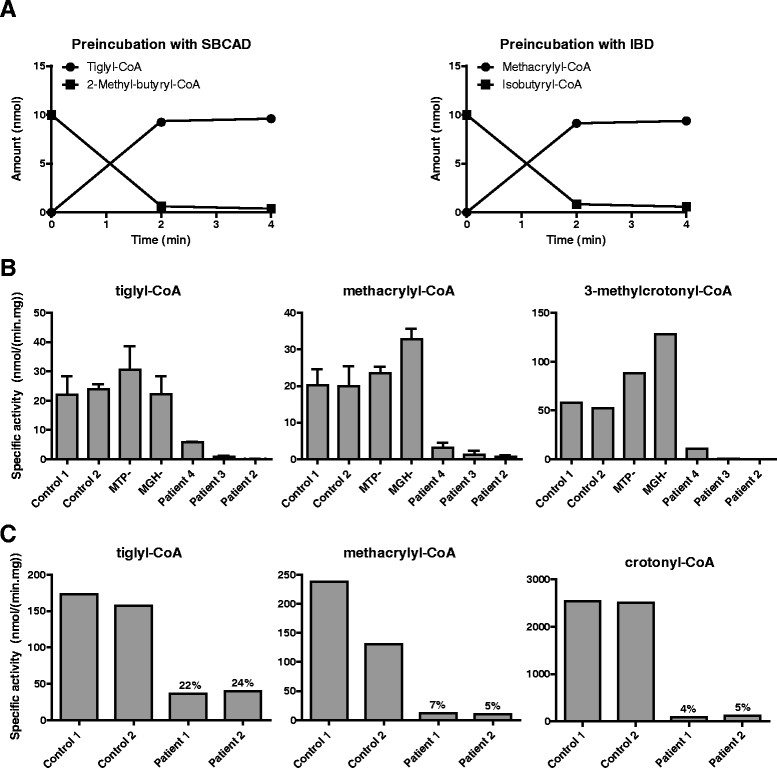
Methods: Metabolite, enzymatic, protein and genetic analyses were performed in four patients, including two siblings. Palmitate loading studies in fibroblasts were performed to study mitochondrial β-oxidation. In addition, enoyl-CoA hydratase activity was measured with crotonyl-CoA, methacrylyl-CoA, tiglyl-CoA and 3-methylcrotonyl-CoA both in fibroblasts and liver to further study the role of SCEH in different metabolic pathways. Analyses of pyruvate dehydrogenase and respiratory chain complexes were performed in multiple tissues of two patients.
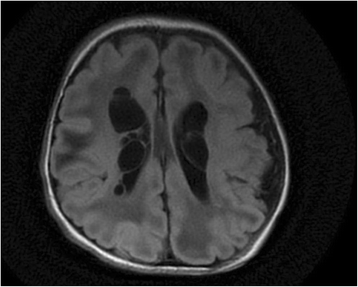
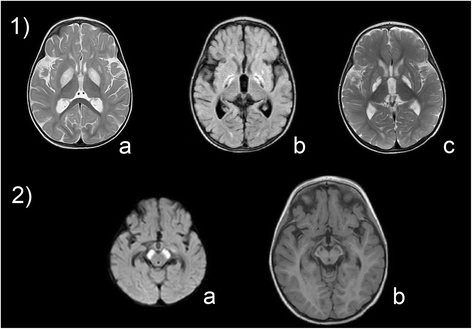
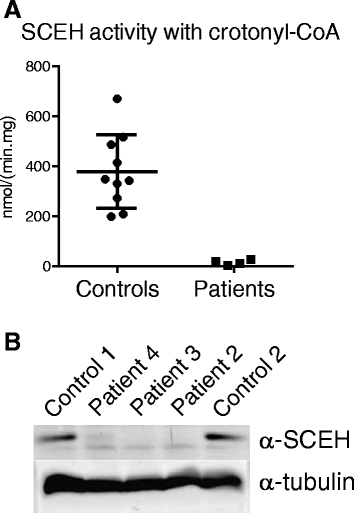
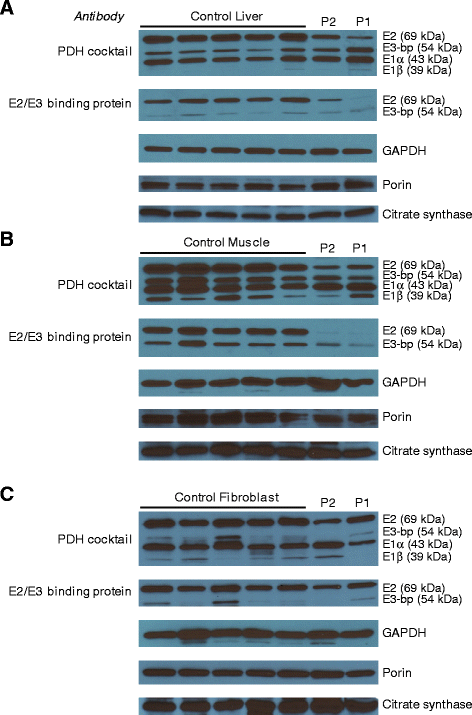
Results: All patients were either homozygous or compound heterozygous for mutations in the ECHS1 gene, had markedly reduced SCEH enzymatic activity and protein level in fibroblasts. All patients presented with lactic acidosis. The first two patients presented with vacuolating leukoencephalopathy and basal ganglia abnormalities. The third patient showed a slow neurodegenerative condition with global brain atrophy and the fourth patient showed Leigh-like lesions with a single episode of metabolic acidosis. Clinical picture and metabolite analysis were not consistent with a mitochondrial fatty acid oxidation disorder, which was supported by the normal palmitate loading test in fibroblasts. Patient fibroblasts displayed deficient hydratase activity with different substrates tested. Pyruvate dehydrogenase activity was markedly reduced in particular in muscle from the most severely affected patients, which was caused by reduced expression of E2 protein, whereas E2 mRNA was increased.
Conclusions: Despite its activity towards substrates from different metabolic pathways, SCEH appears to be only crucial in valine metabolism, but not in isoleucine metabolism, and only of limited importance for mitochondrial fatty acid oxidation. In severely affected patients SCEH deficiency can cause a secondary pyruvate dehydrogenase deficiency contributing to the clinical presentation.
Figures
Similar articles
-
Clinical, biochemical and metabolic characterisation of a mild form of human short-chain enoyl-CoA hydratase deficiency: significance of increased N-acetyl-S-(2-carboxypropyl)cysteine excretion.J Med Genet. 2015 Oct;52(10):691-8. doi: 10.1136/jmedgenet-2015-103231. Epub 2015 Aug 6. J Med Genet. 2015. PMID: 26251176
-
Lethal neonatal case and review of primary short-chain enoyl-CoA hydratase (SCEH) deficiency associated with secondary lymphocyte pyruvate dehydrogenase complex (PDC) deficiency.Mol Genet Metab. 2017 Apr;120(4):342-349. doi: 10.1016/j.ymgme.2017.02.002. Epub 2017 Feb 2. Mol Genet Metab. 2017. PMID: 28202214 Free PMC article. Review.
-
ECHS1 mutations in Leigh disease: a new inborn error of metabolism affecting valine metabolism.Brain. 2014 Nov;137(Pt 11):2903-8. doi: 10.1093/brain/awu216. Epub 2014 Aug 14. Brain. 2014. PMID: 25125611
-
Delineating the neurological phenotype in children with defects in the ECHS1 or HIBCH gene.J Inherit Metab Dis. 2021 Mar;44(2):401-414. doi: 10.1002/jimd.12288. Epub 2020 Aug 16. J Inherit Metab Dis. 2021. PMID: 32677093
-
Clinical, biochemical and metabolic characterization of patients with short-chain enoyl-CoA hydratase(ECHS1) deficiency: two case reports and the review of the literature.BMC Pediatr. 2020 Feb 3;20(1):50. doi: 10.1186/s12887-020-1947-z. BMC Pediatr. 2020. PMID: 32013919 Free PMC article. Review.
Cited by
-
Acyl-CoA dehydrogenase substrate promiscuity: Challenges and opportunities for development of substrate reduction therapy in disorders of valine and isoleucine metabolism.J Inherit Metab Dis. 2023 Sep;46(5):931-942. doi: 10.1002/jimd.12642. Epub 2023 Jun 19. J Inherit Metab Dis. 2023. PMID: 37309295 Free PMC article.
-
Biochemical Markers for the Diagnosis of Mitochondrial Fatty Acid Oxidation Diseases.J Clin Med. 2021 Oct 22;10(21):4855. doi: 10.3390/jcm10214855. J Clin Med. 2021. PMID: 34768374 Free PMC article. Review.
-
Exercise improves choroid plexus epithelial cells metabolism to prevent glial cell-associated neurodegeneration.Front Pharmacol. 2022 Sep 16;13:1010785. doi: 10.3389/fphar.2022.1010785. eCollection 2022. Front Pharmacol. 2022. PMID: 36188600 Free PMC article.
-
ECHS1 deficiency-associated paroxysmal exercise-induced dyskinesias: case presentation and initial benefit of intervention.J Neurol. 2017 Jan;264(1):185-187. doi: 10.1007/s00415-016-8381-z. Epub 2016 Dec 30. J Neurol. 2017. PMID: 28039521
-
Exploring triheptanoin as treatment for short chain enoyl CoA hydratase deficiency.Ann Clin Transl Neurol. 2021 May;8(5):1151-1157. doi: 10.1002/acn3.51359. Epub 2021 May 1. Ann Clin Transl Neurol. 2021. PMID: 33931985 Free PMC article.
References
-
- Fong JC, Schulz H. Purification and properties of pig heart crotonase and the presence of short chain and long chain enoyl coenzyme A hydratases in pig and guinea pig tissues. J Biol Chem. 1977;252:542–7. - PubMed
-
- Shimomura Y, Murakami T, Fujitsuka N, Nakai N, Sato Y, Sugiyama S, et al. Purification and partial characterization of 3-hydroxyisobutyryl-coenzyme A hydrolase of rat liver. J Biol Chem. 1994;269:14248–53. - PubMed
-
- Stern JR, Del Campillo A. Enzymes of fatty acid metabolism. II. Properties of crystalline crotonase. J Biol Chem. 1956;218:985–1002. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

