Profiling and targeting HER2-positive breast cancer using trastuzumab emtansine
- PMID: 25378946
- PMCID: PMC4207068
- DOI: 10.2147/PGPM.S47524
Profiling and targeting HER2-positive breast cancer using trastuzumab emtansine
Abstract
Purpose: This article reviews the mechanism of action of trastuzumab emtansine (T-DM1), existing clinical data relating to its use for human growth factor receptor 2 (HER2)-positive breast cancer, potential pathways of resistance, and ongoing studies evaluating this novel agent.
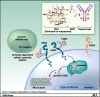
Background: The development of HER2-targeted therapies has dramatically improved clinical outcomes for patients with any stage of HER2-positive breast cancer. Although the positive effect of these treatments cannot be overstated, treatment resistance develops in the vast majority of those diagnosed with stage IV HER2-positive breast cancer. Moreover, HER2-directed therapies are most effective when combined with cytotoxic chemotherapy. The need for chemotherapy leads to significant adverse effects and a clear decrease in quality of life for those dealing with a chronic incurable disease. T-DM1 is a recently developed, novel antibody-drug conjugate in which highly potent maytanisinoid chemotherapy is stably linked to the HER2-targeted monoclonal antibody, trastuzumab.
Results: Preclinical and phase 1-3 clinical data support the significant antitumor activity of T-DM1. Importantly, several randomized studies also now demonstrate its clear superiority in terms of tolerability compared with standard chemotherapy-containing regimens. Its role in the treatment of trastuzumab-resistant metastatic breast cancer has now been established on the basis of the results of two phase 3 randomized studies, EMILIA (An Open-label Study of Trastuzumab Emtansine (T-DM1) vs Capecitabine + Lapatinib in Patients With HER2-positive Locally Advanced or Metastatic Breast Cancer) and TH3RESA (A Study of Trastuzumab Emtansine in Comparison With Treatment of Physician's Choice in Patients With HER2-positive Breast Cancer Who Have Received at Least Two Prior Regimens of HER2-directed Therapy). The most common toxicities seen with T-DM1 are thrombocytopenia and an elevation in liver transaminases. Significant cardiac toxicity has not been demonstrated. Both in vitro cell line-based studies as well as exploratory analyses of archived tumor samples from the clinical trials are seeking to understand potential mechanisms of resistance to T-DM1. Ongoing studies are also evaluating the use of T-DM1 in the first-line metastatic, neoadjuvant, and adjuvant settings, as well as in combination with other targeted therapies.
Conclusion: T-DM1 represents the first successfully developed antibody drug conjugate for the treatment of HER2-positive advanced breast cancer.
Keywords: HER2; Kadcyla; T-DM1; ado-trastuzumab emtansine; metastatic breast cancer; trastuzumab emtansine.
Figures
Similar articles
-
Ado-trastuzumab emtansine: a HER2-positive targeted antibody-drug conjugate.Ann Pharmacother. 2014 Nov;48(11):1484-93. doi: 10.1177/1060028014545354. Epub 2014 Jul 31. Ann Pharmacother. 2014. PMID: 25082874 Review.
-
Primary results of ELAINA: a randomized, multicenter, open-label, phase III study of the efficacy and safety of trastuzumab emtansine vs. lapatinib plus capecitabine in Chinese patients with HER2-positive locally advanced or metastatic breast cancer who have received prior trastuzumab-based therapy.Transl Breast Cancer Res. 2023 Jan 31;4:3. doi: 10.21037/tbcr-23-2. eCollection 2023. Transl Breast Cancer Res. 2023. PMID: 38751488 Free PMC article.
-
Predicting Thrombocytopenia in Patients With Breast Cancer Treated With Ado-trastuzumab Emtansine.Clin Breast Cancer. 2020 Apr;20(2):e220-e228. doi: 10.1016/j.clbc.2019.10.001. Epub 2019 Dec 7. Clin Breast Cancer. 2020. PMID: 31892489
-
Trastuzumab emtansine versus treatment of physician's choice for pretreated HER2-positive advanced breast cancer (TH3RESA): a randomised, open-label, phase 3 trial.Lancet Oncol. 2014 Jun;15(7):689-99. doi: 10.1016/S1470-2045(14)70178-0. Epub 2014 May 2. Lancet Oncol. 2014. PMID: 24793816 Clinical Trial.
-
Trastuzumab Emtansine for Treating HER2-Positive, Unresectable, Locally Advanced or Metastatic Breast Cancer After Treatment with Trastuzumab and a Taxane: An Evidence Review Group Perspective of a NICE Single Technology Appraisal.Pharmacoeconomics. 2016 Jul;34(7):673-80. doi: 10.1007/s40273-016-0386-z. Pharmacoeconomics. 2016. PMID: 26892972 Review.
Cited by
-
The prognostic role of mTOR and p-mTOR for survival in non-small cell lung cancer: a systematic review and meta-analysis.PLoS One. 2015 Feb 13;10(2):e0116771. doi: 10.1371/journal.pone.0116771. eCollection 2015. PLoS One. 2015. PMID: 25680114 Free PMC article. Review.
-
Tribody [(HER2)2xCD16] Is More Effective Than Trastuzumab in Enhancing γδ T Cell and Natural Killer Cell Cytotoxicity Against HER2-Expressing Cancer Cells.Front Immunol. 2018 Apr 19;9:814. doi: 10.3389/fimmu.2018.00814. eCollection 2018. Front Immunol. 2018. PMID: 29725336 Free PMC article.
-
Prognostic value of programmed cell death-ligand 1 expression in patients with non-small-cell lung cancer: evidence from an updated meta-analysis.Onco Targets Ther. 2015 Dec 1;8:3595-601. doi: 10.2147/OTT.S91469. eCollection 2015. Onco Targets Ther. 2015. PMID: 26664143 Free PMC article.
-
From 3D spheroids to tumor bearing mice: efficacy and distribution studies of trastuzumab-docetaxel immunoliposome in breast cancer.Int J Nanomedicine. 2018 Oct 23;13:6677-6688. doi: 10.2147/IJN.S179290. eCollection 2018. Int J Nanomedicine. 2018. PMID: 30425482 Free PMC article.
References
-
- Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235(4785):177–182. - PubMed
-
- Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344(11):783–792. - PubMed
-
- Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353(16):1673–1684. - PubMed
-
- Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Herceptin Adjuvant (HERA) Trial Study Team Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353(16):1659–1672. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

