Fatty acids from membrane lipids become incorporated into lipid bodies during Myxococcus xanthus differentiation
- PMID: 24906161
- PMCID: PMC4048283
- DOI: 10.1371/journal.pone.0099622
Fatty acids from membrane lipids become incorporated into lipid bodies during Myxococcus xanthus differentiation
Abstract
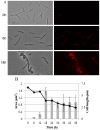
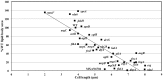
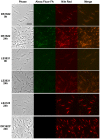
Myxococcus xanthus responds to amino acid limitation by producing fruiting bodies containing dormant spores. During development, cells produce triacylglycerides in lipid bodies that become consumed during spore maturation. As the cells are starved to induce development, the production of triglycerides represents a counterintuitive metabolic switch. In this paper, lipid bodies were quantified in wild-type strain DK1622 and 33 developmental mutants at the cellular level by measuring the cross sectional area of the cell stained with the lipophilic dye Nile red. We provide five lines of evidence that triacylglycerides are derived from membrane phospholipids as cells shorten in length and then differentiate into myxospores. First, in wild type cells, lipid bodies appear early in development and their size increases concurrent with an 87% decline in membrane surface area. Second, developmental mutants blocked at different stages of shortening and differentiation accumulated lipid bodies proportionate with their cell length with a Pearson's correlation coefficient of 0.76. Third, peripheral rods, developing cells that do not produce lipid bodies, fail to shorten. Fourth, genes for fatty acid synthesis are down-regulated while genes for fatty acid degradation are up regulated. Finally, direct movement of fatty acids from membrane lipids in growing cells to lipid bodies in developing cells was observed by pulse labeling cells with palmitate. Recycling of lipids released by Programmed Cell Death appears not to be necessary for lipid body production as a fadL mutant was defective in fatty acid uptake but proficient in lipid body production. The lipid body regulon involves many developmental genes that are not specifically involved in fatty acid synthesis or degradation. MazF RNA interferase and its target, enhancer-binding protein Nla6, appear to negatively regulate cell shortening and TAG accumulation whereas most cell-cell signals activate these processes.
Conflict of interest statement
Figures
Similar articles
-
Fatty Acid Oxidation Is Required for Myxococcus xanthus Development.J Bacteriol. 2018 Apr 24;200(10):e00572-17. doi: 10.1128/JB.00572-17. Print 2018 May 15. J Bacteriol. 2018. PMID: 29507089 Free PMC article.
-
Two lipid signals guide fruiting body development of Myxococcus xanthus.mBio. 2014 Feb 11;5(1):e00939-13. doi: 10.1128/mBio.00939-13. mBio. 2014. PMID: 24520059 Free PMC article.
-
Neutral and Phospholipids of the Myxococcus xanthus Lipodome during Fruiting Body Formation and Germination.Appl Environ Microbiol. 2015 Oct;81(19):6538-47. doi: 10.1128/AEM.01537-15. Epub 2015 Jul 10. Appl Environ Microbiol. 2015. PMID: 26162876 Free PMC article.
-
Branched-chain fatty acids: the case for a novel form of cell-cell signalling during Myxococcus xanthus development.Mol Microbiol. 1995 Apr;16(2):171-5. doi: 10.1111/j.1365-2958.1995.tb02290.x. Mol Microbiol. 1995. PMID: 7565080 Review.
-
Genetics of gliding motility and development in Myxococcus xanthus.Arch Microbiol. 1995 Nov;164(5):309-23. doi: 10.1007/BF02529977. Arch Microbiol. 1995. PMID: 8572884 Review.
Cited by
-
Short-range C-signaling restricts cheating behavior during Myxococcus xanthus development.mBio. 2024 Nov 13;15(11):e0244024. doi: 10.1128/mbio.02440-24. Epub 2024 Oct 18. mBio. 2024. PMID: 39422488 Free PMC article.
-
Proteome Analyses of Soil Bacteria Grown in the Presence of Potato Suberin, a Recalcitrant Biopolymer.Microbes Environ. 2016 Dec 23;31(4):418-426. doi: 10.1264/jsme2.ME15195. Epub 2016 Oct 28. Microbes Environ. 2016. PMID: 27795492 Free PMC article.
-
Cell-cell transfer of adaptation traits benefits kin and actor in a cooperative microbe.Proc Natl Acad Sci U S A. 2024 Jul 23;121(30):e2402559121. doi: 10.1073/pnas.2402559121. Epub 2024 Jul 16. Proc Natl Acad Sci U S A. 2024. PMID: 39012831 Free PMC article.
-
Myxococcus CsgA, Drosophila Sniffer, and human HSD10 are cardiolipin phospholipases.Genes Dev. 2015 Sep 15;29(18):1903-14. doi: 10.1101/gad.268482.115. Epub 2015 Sep 3. Genes Dev. 2015. PMID: 26338420 Free PMC article.
-
Quantification of Myxococcus xanthus Aggregation and Rippling Behaviors: Deep-Learning Transformation of Phase-Contrast into Fluorescence Microscopy Images.Microorganisms. 2021 Sep 14;9(9):1954. doi: 10.3390/microorganisms9091954. Microorganisms. 2021. PMID: 34576849 Free PMC article.
References
-
- Alvarez HM, Steinbuchel A (2002) Triacylglycerols in prokaryotic microorganisms. Appl Microbiol Biotechnol 60: 367–376. - PubMed
-
- Waltermann M, Hinz A, Robenek H, Troyer D, Reichelt R, et al. (2005) Mechanism of lipid-body formation in prokaryotes: how bacteria fatten up. Mol Microbiol 55: 750–763. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

