Dysfunction of collagen synthesis and secretion in chondrocytes induced by wisp3 mutation
- PMID: 23573089
- PMCID: PMC3614060
- DOI: 10.1155/2013/679763
Dysfunction of collagen synthesis and secretion in chondrocytes induced by wisp3 mutation
Abstract
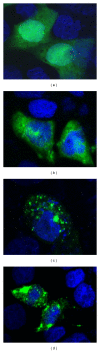
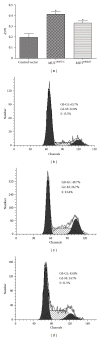
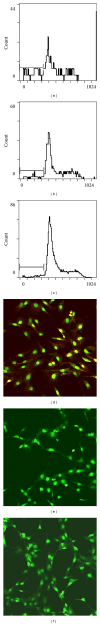
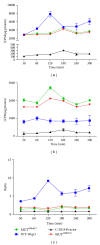
Wisp3 gene mutation was shown to cause spondyloepiphyseal dysplasia tarda with progressive arthropathy (SRDT-PA), but the underlying mechanism is not clear. To clarify this mechanism, we constructed the wild and mutated Wisp3 expression vectors and transfected into human chondrocytes lines C-20/A4; Wisp3 proteins subcellular localization, cell proliferation, cell apoptosis, and Wisp3-mediated gene expression were determined, and dynamic secretion of collagen in transfected chondrocytes was analyzed by (14)C-proline incorporation experiment. Mutated Wisp3 protein increased proliferation activity, decreased apoptosis of C-20/A4 cells, and aggregated abnormally in cytoplasm. Expression of collagen II was also downregulated in C-20/A4 cells transfected with mutated Wisp3. Wild type Wisp3 transfection increased intracellular collagen content and extracellular collagen secretion, but the mutated Wisp3 lost this function, and the peak phase of collagen secretion was delayed in mutated Wisp3 transfected cells. Thus abnormal protein distribution, cell proliferation, collagen synthesis, and secretion in Wisp3 mutated chondrocytes might contribute to the pathogenesis of SEDT-PA.
Figures


Similar articles
-
Progressive pseudorheumatoid dysplasia: a rare childhood disease.Rheumatol Int. 2019 Mar;39(3):441-452. doi: 10.1007/s00296-018-4170-6. Epub 2018 Oct 16. Rheumatol Int. 2019. PMID: 30327864 Review.
-
Mutant WISP3 triggers the phenotype shift of articular chondrocytes by promoting sensitivity to IGF-1 hypothesis of spondyloepiphyseal dysplasia tarda with progressive arthropathy (SEDT-PA).Med Hypotheses. 2007;68(6):1406-10. doi: 10.1016/j.mehy.2006.06.046. Epub 2007 Mar 23. Med Hypotheses. 2007. PMID: 17363178
-
[Construction of WISP3 gene's mutants in SEDT-PA and their expression in COS-7 cells].Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2008 Jan;33(1):8-15. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2008. PMID: 18245897 Chinese.
-
WISP3 suppresses insulin-like growth factor signaling in human chondrocytes.Mol Cell Endocrinol. 2007 Dec 15;279(1-2):1-8. doi: 10.1016/j.mce.2007.08.007. Epub 2007 Aug 28. Mol Cell Endocrinol. 2007. PMID: 17942216
-
The diagnostic challenge of progressive pseudorheumatoid dysplasia (PPRD): a review of clinical features, radiographic features, and WISP3 mutations in 63 affected individuals.Am J Med Genet C Semin Med Genet. 2012 Aug 15;160C(3):217-29. doi: 10.1002/ajmg.c.31333. Epub 2012 Jul 12. Am J Med Genet C Semin Med Genet. 2012. PMID: 22791401 Review.
Cited by
-
Case Report: Recurrent Variant c.298 TA in CCN6 Gene Found in Progressive Pseudorheumatoid Dysplasia Patients From Patni Community of Gujarat: A Report of Three Cases.Front Genet. 2021 Sep 28;12:724824. doi: 10.3389/fgene.2021.724824. eCollection 2021. Front Genet. 2021. PMID: 34650595 Free PMC article.
-
Progressive pseudorheumatoid dysplasia: a rare childhood disease.Rheumatol Int. 2019 Mar;39(3):441-452. doi: 10.1007/s00296-018-4170-6. Epub 2018 Oct 16. Rheumatol Int. 2019. PMID: 30327864 Review.
-
Mesenchymal stromal cells from a progressive pseudorheumatoid dysplasia patient show altered osteogenic differentiation.Eur J Med Res. 2022 Apr 25;27(1):57. doi: 10.1186/s40001-022-00683-2. Eur J Med Res. 2022. PMID: 35462544 Free PMC article.
-
Novel COL2A1 variant (c.619G>A, p.Gly207Arg) manifesting as a phenotype similar to progressive pseudorheumatoid dysplasia and spondyloepiphyseal dysplasia, Stanescu type.Hum Mutat. 2015 Oct;36(10):1004-8. doi: 10.1002/humu.22839. Epub 2015 Aug 6. Hum Mutat. 2015. PMID: 26183434 Free PMC article.
-
CCN6 mutation detection in Chinese patients with progressive pseudo-rheumatoid dysplasia and identification of four novel mutations.Mol Genet Genomic Med. 2020 Jul;8(7):e1261. doi: 10.1002/mgg3.1261. Epub 2020 Apr 29. Mol Genet Genomic Med. 2020. PMID: 32351055 Free PMC article.
References
-
- Hurvitz JR, Suwairi WM, Van Hul W, et al. Mutations in the CCN gene family member WISP3 cause progressive pseudorheumatoid dysplasia. Nature Genetics. 1999;23(1):94–98. - PubMed
-
- Perbal B, Martinerie C, Sainson R, Werner M, He B, Roizman B. The C-terminal domain of the regulatory protein NOVH is sufficient to promote interaction with fibulin 1C: a clue for a role of NOVH in cell-adhesion signaling. Proceedings of the National Academy of Sciences of the United States of America. 1999;96(3):869–874. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

