Should a hospitalized child receive empiric treatment with acyclovir?
- PMID: 23244394
- PMCID: PMC3541113
- DOI: 10.1186/1824-7288-38-72
Should a hospitalized child receive empiric treatment with acyclovir?
Abstract
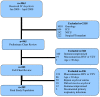
Background: Herpes simplex encephalitis is associated with substantial morbidity and mortality and may be related to timely diagnosis and treatment. While awaiting the results of testing, hospitalization and empiric treatment with acyclovir is recommended, though the direct and indirect costs associated with this management are substantial. We sought to examine children hospitalized for possible herpes simplex encephalitis, following clinical and laboratory assessment in the emergency department, and empiric treatment with acyclovir, in order to describe the proportion receiving a complete course of treatment; and to identify the clinical variables which are associated with receiving a complete course, as compared with an incomplete course of acyclovir.
Methods: Hospitalized children prescribed acyclovir were included in this case control study. Clinical, laboratory and diagnostic variables were abstracted for children prescribed a complete (≥ 14 days) or an incomplete course (<14 days) of acyclovir. Odds ratios and 95% confidence intervals were calculated.
Results: 289 children met eligibility criteria, 30 (10%) received a complete course and 259 (90%) received an incomplete course. A history of mucocutaneous herpes simplex virus infection (p < 0.01), Glasgow Coma Scale ≤ 13 (p = 0.02), focal neurologic findings (p = 0.001) and elevated cerebrospinal fluid white blood cell count (p = 0.05) were associated with a complete course of acyclovir.
Conclusions: Many children did not complete a full course of therapy. Unnecessary testing and treatment is burdensome to families and the health care system. Possible predictive variables include abnormal Glascow Coma Scale, focal neurologic findings and cerebrospinal fluid pleocytosis.
Figures
Similar articles
-
Clinical outcomes in children with herpes simplex encephalitis receiving steroid therapy.J Clin Virol. 2016 Jul;80:87-92. doi: 10.1016/j.jcv.2016.05.002. Epub 2016 May 11. J Clin Virol. 2016. PMID: 27218417
-
Impact of a Rapid Herpes Simplex Virus PCR Assay on Duration of Acyclovir Therapy.J Clin Microbiol. 2017 May;55(5):1557-1565. doi: 10.1128/JCM.02559-16. Epub 2017 Mar 8. J Clin Microbiol. 2017. PMID: 28275080 Free PMC article.
-
Relapsing Herpes simplex virus encephalitis despite high-dose acyclovir therapy: a case report.Turk J Pediatr. 2008 Jul-Aug;50(4):380-2. Turk J Pediatr. 2008. PMID: 19014054
-
Herpes simplex virus-1 encephalitis: a review of current disease management with three case reports.Antivir Chem Chemother. 2012 Sep 25;23(1):13-8. doi: 10.3851/IMP2129. Antivir Chem Chemother. 2012. PMID: 23018202 Review.
-
Herpes simplex encephalitis : from virus to therapy.Infect Disord Drug Targets. 2011 Jun;11(3):235-50. doi: 10.2174/187152611795768088. Infect Disord Drug Targets. 2011. PMID: 21488834 Review.
Cited by
-
Update on Viral Infections Involving the Central Nervous System in Pediatric Patients.Children (Basel). 2021 Sep 6;8(9):782. doi: 10.3390/children8090782. Children (Basel). 2021. PMID: 34572214 Free PMC article. Review.
-
Encephalitis diagnosis using metagenomics: application of next generation sequencing for undiagnosed cases.J Infect. 2018 Mar;76(3):225-240. doi: 10.1016/j.jinf.2017.12.014. Epub 2018 Jan 2. J Infect. 2018. PMID: 29305150 Free PMC article. Review.
References
-
- Elbers JM, Bitnun A, Richardson SE, Ford-Jones EL, Tellier R, Wald RM, Petric M, Kolski H, Heurter H, MacGregor D. A 12-year prospective study of childhood herpes simplex encephalitis: is there a broader spectrum of disease? Pediatrics. 2007;119:e399–e407. doi: 10.1542/peds.2006-1494. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous