Agammaglobulinemia and absent B lineage cells in a patient lacking the p85α subunit of PI3K
- PMID: 22351933
- PMCID: PMC3302225
- DOI: 10.1084/jem.20112533
Agammaglobulinemia and absent B lineage cells in a patient lacking the p85α subunit of PI3K
Abstract
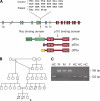
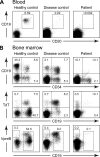
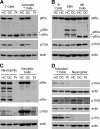
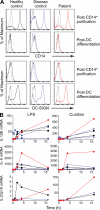
Whole exome sequencing was used to determine the causative gene in patients with B cell defects of unknown etiology. A homozygous premature stop codon in exon 6 of PIK3R1 was identified in a young woman with colitis and absent B cells. The mutation results in the absence of p85α but normal expression of the p50α and p55α regulatory subunits of PI3K. Bone marrow aspirates from the patient showed <0.1% CD19(+) B cells with normal percentages of TdT(+)VpreB(+)CD19(-) B cell precursors. This developmental block is earlier than that seen in patients with defects in the B cell receptor signaling pathway or in a strain of engineered mice with a similar defect in p85α. The number and function of the patient's T cells were normal. However, Western blot showed markedly decreased p110δ, as well as absent p85α, in patient T cells, neutrophils, and dendritic cells. The patient had normal growth and development and normal fasting glucose and insulin. Mice with p85α deficiency have insulin hypersensitivity, defective platelet function, and abnormal mast cell development. In contrast, the absence of p85α in the patient results in an early and severe defect in B cell development but minimal findings in other organ systems.
Figures
Similar articles
-
Divergent roles of the regulatory subunits of class IA PI3K.Front Endocrinol (Lausanne). 2024 Jan 22;14:1152579. doi: 10.3389/fendo.2023.1152579. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38317714 Free PMC article. Review.
-
Reduced T cell expansion by a superantigen as a result of impaired B cell development in mice deficient for the p85alpha regulatory subunit of PI3K.J Leukoc Biol. 2010 Mar;87(3):493-500. doi: 10.1189/jlb.0708440. Epub 2009 Dec 9. J Leukoc Biol. 2010. PMID: 20007249
-
Mutation analysis of the Bruton's tyrosine kinase gene in X-linked agammaglobulinemia: identification of a mutation which affects the same codon as is altered in immunodeficient xid mice.Hum Mol Genet. 1994 Jan;3(1):161-6. doi: 10.1093/hmg/3.1.161. Hum Mol Genet. 1994. PMID: 8162018
-
Class I(A) PI3Kinase regulatory subunit, p85α, mediates mast cell development through regulation of growth and survival related genes.PLoS One. 2012;7(1):e28979. doi: 10.1371/journal.pone.0028979. Epub 2012 Jan 4. PLoS One. 2012. PMID: 22238586 Free PMC article.
-
PIK3R1 Mutation Associated with Hyper IgM (APDS2 Syndrome): A Case Report and Review of the Literature.Endocr Metab Immune Disord Drug Targets. 2019;19(7):941-958. doi: 10.2174/1871530319666190225114739. Endocr Metab Immune Disord Drug Targets. 2019. PMID: 30799802 Review.
Cited by
-
Newborn screening for SCID identifies patients with ataxia telangiectasia.J Clin Immunol. 2013 Apr;33(3):540-9. doi: 10.1007/s10875-012-9846-1. Epub 2012 Dec 20. J Clin Immunol. 2013. PMID: 23264026 Free PMC article.
-
ICON: the early diagnosis of congenital immunodeficiencies.J Clin Immunol. 2014 May;34(4):398-424. doi: 10.1007/s10875-014-0003-x. Epub 2014 Mar 12. J Clin Immunol. 2014. PMID: 24619621 Review.
-
A recurrent dominant negative E47 mutation causes agammaglobulinemia and BCR(-) B cells.J Clin Invest. 2013 Nov;123(11):4781-5. doi: 10.1172/JCI71927. J Clin Invest. 2013. PMID: 24216514 Free PMC article.
-
The diagnostic approach to monogenic very early onset inflammatory bowel disease.Gastroenterology. 2014 Nov;147(5):990-1007.e3. doi: 10.1053/j.gastro.2014.07.023. Epub 2014 Jul 21. Gastroenterology. 2014. PMID: 25058236 Free PMC article. Review.
-
PI3K Isoforms in B Cells.Curr Top Microbiol Immunol. 2022;436:235-254. doi: 10.1007/978-3-031-06566-8_10. Curr Top Microbiol Immunol. 2022. PMID: 36243847
References
-
- Aiuti A., Tavian M., Cipponi A., Ficara F., Zappone E., Hoxie J., Peault B., Bordignon C. 1999. Expression of CXCR4, the receptor for stromal cell-derived factor-1 on fetal and adult human lympho-hematopoietic progenitors. Eur. J. Immunol. 29:1823–1831 10.1002/(SICI)1521-4141(199906)29:06<1823::AID-IMMU1823>3.0.CO;2-B - DOI - PubMed
-
- Brown J.B., Cheresh P., Goretsky T., Managlia E., Grimm G.R., Ryu H., Zadeh M., Dirisina R., Barrett T.A. 2011. Epithelial phosphatidylinositol-3-kinase signaling is required for β-catenin activation and host defense against Citrobacter rodentium infection. Infect. Immun. 79:1863–1872 10.1128/IAI.01025-10 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

