Effects of pathogenic proline mutations on myosin assembly
- PMID: 22155079
- PMCID: PMC3267876
- DOI: 10.1016/j.jmb.2011.11.042
Effects of pathogenic proline mutations on myosin assembly
Abstract
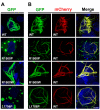
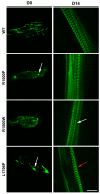
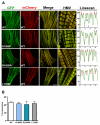
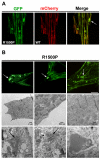
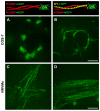
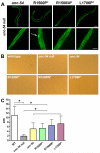
Laing distal myopathy (MPD1) is a genetically dominant myopathy characterized by early and selective weakness of the distal muscles. Mutations in the MYH7 gene encoding for the β-myosin heavy chain are the underlying genetic cause of MPD1. However, their pathogenic mechanisms are currently unknown. Here, we measure the biological effects of the R1500P and L1706P MPD1 mutations in different cellular systems. We show that, while the two mutations inhibit myosin self-assembly in non-muscle cells, they do not prevent incorporation of the mutant myosin into sarcomeres. Nevertheless, we find that the L1706P mutation affects proper antiparallel myosin association by accumulating in the bare zone of the sarcomere. Furthermore, bimolecular fluorescence complementation assay shows that the α-helix containing the R1500P mutation folds into homodimeric (mutant/mutant) and heterodimeric [mutant/wild type (WT)] myosin molecules that are competent for sarcomere incorporation. Both mutations also form aggregates consisting of cytoplasmic vacuoles surrounding paracrystalline arrays and amorphous rod-like inclusions that sequester WT myosin. Myosin aggregates were also detected in transgenic nematodes expressing the R1500P mutation. By showing that the two MPD1 mutations can have dominant effects on distinct components of the contractile apparatus, our data provide the first insights into the pathogenesis of the disease.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Myosinopathies: pathology and mechanisms.Acta Neuropathol. 2013 Jan;125(1):3-18. doi: 10.1007/s00401-012-1024-2. Epub 2012 Aug 5. Acta Neuropathol. 2013. PMID: 22918376 Free PMC article. Review.
-
Two novel MYH7 proline substitutions cause Laing Distal Myopathy-like phenotypes with variable expressivity and neck extensor contracture.BMC Med Genet. 2016 Aug 12;17(1):57. doi: 10.1186/s12881-016-0315-1. BMC Med Genet. 2016. PMID: 27519903 Free PMC article.
-
A Laing distal myopathy-associated proline substitution in the β-myosin rod perturbs myosin cross-bridging activity.J Clin Invest. 2024 May 1;134(9):e172599. doi: 10.1172/JCI172599. J Clin Invest. 2024. PMID: 38690726 Free PMC article.
-
A1603P and K1617del, Mutations in β-Cardiac Myosin Heavy Chain that Cause Laing Early-Onset Distal Myopathy, Affect Secondary Structure and Filament Formation In Vitro and In Vivo.J Mol Biol. 2018 May 11;430(10):1459-1478. doi: 10.1016/j.jmb.2018.04.006. Epub 2018 Apr 14. J Mol Biol. 2018. PMID: 29660325 Free PMC article.
-
Hereditary myosin myopathies.Neuromuscul Disord. 2007 May;17(5):355-67. doi: 10.1016/j.nmd.2007.02.008. Epub 2007 Apr 16. Neuromuscul Disord. 2007. PMID: 17434305 Review.
Cited by
-
Novel mutations widen the phenotypic spectrum of slow skeletal/β-cardiac myosin (MYH7) distal myopathy.Hum Mutat. 2014 Jul;35(7):868-79. doi: 10.1002/humu.22553. Epub 2014 May 21. Hum Mutat. 2014. PMID: 24664454 Free PMC article.
-
Myosinopathies: pathology and mechanisms.Acta Neuropathol. 2013 Jan;125(1):3-18. doi: 10.1007/s00401-012-1024-2. Epub 2012 Aug 5. Acta Neuropathol. 2013. PMID: 22918376 Free PMC article. Review.
-
Skip residues modulate the structural properties of the myosin rod and guide thick filament assembly.Proc Natl Acad Sci U S A. 2015 Jul 21;112(29):E3806-15. doi: 10.1073/pnas.1505813112. Epub 2015 Jul 6. Proc Natl Acad Sci U S A. 2015. PMID: 26150528 Free PMC article.
-
Two novel MYH7 proline substitutions cause Laing Distal Myopathy-like phenotypes with variable expressivity and neck extensor contracture.BMC Med Genet. 2016 Aug 12;17(1):57. doi: 10.1186/s12881-016-0315-1. BMC Med Genet. 2016. PMID: 27519903 Free PMC article.
-
Design considerations in coiled-coil fusion constructs for the structural determination of a problematic region of the human cardiac myosin rod.J Struct Biol. 2017 Dec;200(3):219-228. doi: 10.1016/j.jsb.2017.07.006. Epub 2017 Jul 22. J Struct Biol. 2017. PMID: 28743637 Free PMC article.
References
-
- Mastaglia FL, Phillips BA, Cala LA, Meredith C, Egli S, Akkari PA, Laing NG. Early onset chromosome 14-linked distal myopathy (Laing) Neuromuscul Disord. 2002;12:350–7. - PubMed
-
- Muelas N, Hackman P, Luque H, Garces-Sanchez M, Azorin I, Suominen T, Sevilla T, Mayordomo F, Gomez L, Marti P, Millan J. Maria, Udd B, Vilchez JJ. MYH7 gene tail mutation causing myopathic profiles beyond Laing distal myopathy. Neurology. 2010;75:732–41. - PubMed
-
- Craig R, Woodhead JL. Structure and function of myosin filaments. Curr Opin Struct Biol. 2006;16:204–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

