Atypical hemolytic uremic syndrome
- PMID: 21902819
- PMCID: PMC3198674
- DOI: 10.1186/1750-1172-6-60
Atypical hemolytic uremic syndrome
Abstract
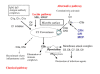
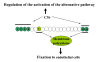
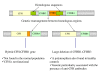
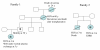
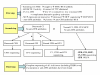
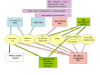
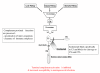
Hemolytic uremic syndrome (HUS) is defined by the triad of mechanical hemolytic anemia, thrombocytopenia and renal impairment. Atypical HUS (aHUS) defines non Shiga-toxin-HUS and even if some authors include secondary aHUS due to Streptococcus pneumoniae or other causes, aHUS designates a primary disease due to a disorder in complement alternative pathway regulation. Atypical HUS represents 5 -10% of HUS in children, but the majority of HUS in adults. The incidence of complement-aHUS is not known precisely. However, more than 1000 aHUS patients investigated for complement abnormalities have been reported. Onset is from the neonatal period to the adult age. Most patients present with hemolytic anemia, thrombocytopenia and renal failure and 20% have extra renal manifestations. Two to 10% die and one third progress to end-stage renal failure at first episode. Half of patients have relapses. Mutations in the genes encoding complement regulatory proteins factor H, membrane cofactor protein (MCP), factor I or thrombomodulin have been demonstrated in 20-30%, 5-15%, 4-10% and 3-5% of patients respectively, and mutations in the genes of C3 convertase proteins, C3 and factor B, in 2-10% and 1-4%. In addition, 6-10% of patients have anti-factor H antibodies. Diagnosis of aHUS relies on 1) No associated disease 2) No criteria for Shigatoxin-HUS (stool culture and PCR for Shiga-toxins; serology for anti-lipopolysaccharides antibodies) 3) No criteria for thrombotic thrombocytopenic purpura (serum ADAMTS 13 activity > 10%). Investigation of the complement system is required (C3, C4, factor H and factor I plasma concentration, MCP expression on leukocytes and anti-factor H antibodies; genetic screening to identify risk factors). The disease is familial in approximately 20% of pedigrees, with an autosomal recessive or dominant mode of transmission. As penetrance of the disease is 50%, genetic counseling is difficult. Plasmatherapy has been first line treatment until presently, without unquestionable demonstration of efficiency. There is a high risk of post-transplant recurrence, except in MCP-HUS. Case reports and two phase II trials show an impressive efficacy of the complement C5 blocker eculizumab, suggesting it will be the next standard of care. Except for patients treated by intensive plasmatherapy or eculizumab, the worst prognosis is in factor H-HUS, as mortality can reach 20% and 50% of survivors do not recover renal function. Half of factor I-HUS progress to end-stage renal failure. Conversely, most patients with MCP-HUS have preserved renal function. Anti-factor H antibodies-HUS has favourable outcome if treated early.
Figures


Similar articles
-
[Pathophysiology of atypical hemolytic uremic syndrome. Ten years of progress, from laboratory to patient].Biol Aujourdhui. 2013;207(4):231-40. doi: 10.1051/jbio/2013027. Epub 2014 Mar 5. Biol Aujourdhui. 2013. PMID: 24594571 Review. French.
-
[Atypical hemolytic-uremic syndrome related to abnormalities within the complement system].Rev Med Interne. 2011 Apr;32(4):232-40. doi: 10.1016/j.revmed.2009.09.039. Epub 2011 Mar 3. Rev Med Interne. 2011. PMID: 21376430 French.
-
Relative role of genetic complement abnormalities in sporadic and familial aHUS and their impact on clinical phenotype.Clin J Am Soc Nephrol. 2010 Oct;5(10):1844-59. doi: 10.2215/CJN.02210310. Epub 2010 Jul 1. Clin J Am Soc Nephrol. 2010. PMID: 20595690 Free PMC article.
-
Genetic and functional analyses of membrane cofactor protein (CD46) mutations in atypical hemolytic uremic syndrome.J Am Soc Nephrol. 2006 Jul;17(7):2017-25. doi: 10.1681/ASN.2005101051. Epub 2006 Jun 8. J Am Soc Nephrol. 2006. PMID: 16762990
-
Atypical hemolytic uremic syndrome.Semin Nephrol. 2013 Nov;33(6):508-30. doi: 10.1016/j.semnephrol.2013.08.003. Semin Nephrol. 2013. PMID: 24161037 Free PMC article. Review.
Cited by
-
Eculizumab therapy for atypical haemolytic uraemic syndrome due to a gain-of-function mutation of complement factor B.Pediatr Nephrol. 2013 Aug;28(8):1315-8. doi: 10.1007/s00467-013-2492-x. Epub 2013 Apr 28. Pediatr Nephrol. 2013. PMID: 23624872
-
Complement factor I deficiency: a not so rare immune defect: characterization of new mutations and the first large gene deletion.Orphanet J Rare Dis. 2012 Jun 18;7:42. doi: 10.1186/1750-1172-7-42. Orphanet J Rare Dis. 2012. PMID: 22710145 Free PMC article.
-
Fecal diagnostics in combination with serology: best test to establish STEC-HUS.Pediatr Nephrol. 2016 Nov;31(11):2163-70. doi: 10.1007/s00467-016-3420-7. Epub 2016 May 30. Pediatr Nephrol. 2016. PMID: 27240858 Free PMC article.
-
COVID-19: A Rare Cause of Hemolytic Uremic Syndrome.Cureus. 2022 Aug 13;14(8):e27962. doi: 10.7759/cureus.27962. eCollection 2022 Aug. Cureus. 2022. PMID: 36120203 Free PMC article.
-
Atypical hemolytic uremic syndrome in a child: A rare case report.Clin Case Rep. 2023 Dec 27;12(1):e8356. doi: 10.1002/ccr3.8356. eCollection 2024 Jan. Clin Case Rep. 2023. PMID: 38161638 Free PMC article.
References
-
- Besbas N, Karpman D, Landau D, Loirat C, Proesmans W, Remuzzi G, Rizzoni G, Taylor CM, Van de Kar N, Zimmerhackl LB. A classification of hemolytic uremic syndrome and thrombotic thrombocytopenic purpura and related disorders. Kidney Int. 2006;70:423–431. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

