Clinical and molecular characterization of a family with a dominant renin gene mutation and response to treatment with fludrocortisone
- PMID: 21084044
- PMCID: PMC4264543
- DOI: 10.5414/cnp74411
Clinical and molecular characterization of a family with a dominant renin gene mutation and response to treatment with fludrocortisone
Abstract
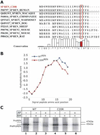
Background: A family was identified with autosomal dominant inheritance of anemia, polyuria, hyperuricemia, and chronic kidney disease. Mutational analysis revealed a novel heterozygous mutation c.58T > C resulting in the amino acid substitution of cysteine for arginine in the preprorenin signal sequence (p.cys20Arg) occurring in all affected members.
Methods: Effects of the identified mutation were characterized using in vitro and in vivo studies. Affected individuals were clinically characterized before and after administration of fludrocortisone.
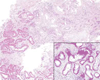
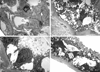
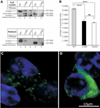
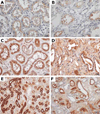
Results: The mutation affects endoplasmic reticulum co-translational translocation and posttranslational processing, resulting in massive accumulation of non-glycosylated preprorenin in the cytoplasm. This affects expression of intra-renal RAS components and leads to ultrastructural damage of the kidney. Affected individuals suffered from anemia, hyperuricemia, decreased urinary concentrating ability, and progressive chronic kidney disease. Treatment with fludrocortisone in an affected 10-year-old child resulted in an increase in blood pressure and estimated glomerular filtration rate.
Conclusions: A novel REN gene mutation resulted in an alteration in the amino acid sequence of the renin signal sequence and caused childhood anemia, polyuria, and kidney disease. Treatment with fludrocortisone improved renal function in an affected child. Nephrologists should consider REN mutational analysis in families with autosomal dominant inheritance of chronic kidney disease, especially if they suffer from anemia, hyperuricemia, and polyuria in childhood.
Figures
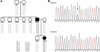

Similar articles
-
Autosomal Dominant Tubulointerstitial Kidney Disease.Adv Chronic Kidney Dis. 2017 Mar;24(2):86-93. doi: 10.1053/j.ackd.2016.11.012. Adv Chronic Kidney Dis. 2017. PMID: 28284384 Free PMC article. Review.
-
Autosomal dominant mutation in the signal peptide of renin in a kindred with anemia, hyperuricemia, and CKD.Am J Kidney Dis. 2011 Nov;58(5):821-5. doi: 10.1053/j.ajkd.2011.06.029. Epub 2011 Sep 8. Am J Kidney Dis. 2011. PMID: 21903317 Free PMC article.
-
Dominant renin gene mutations associated with early-onset hyperuricemia, anemia, and chronic kidney failure.Am J Hum Genet. 2009 Aug;85(2):204-13. doi: 10.1016/j.ajhg.2009.07.010. Epub 2009 Aug 6. Am J Hum Genet. 2009. PMID: 19664745 Free PMC article.
-
Heterozygous Loss-of-Function SEC61A1 Mutations Cause Autosomal-Dominant Tubulo-Interstitial and Glomerulocystic Kidney Disease with Anemia.Am J Hum Genet. 2016 Jul 7;99(1):174-87. doi: 10.1016/j.ajhg.2016.05.028. Am J Hum Genet. 2016. PMID: 27392076 Free PMC article.
-
Autosomal Dominant Tubulointerstitial Kidney Disease Due to UMOD Mutation: A Two-Case Report and Literature Review.Nephron. 2019;143(4):282-287. doi: 10.1159/000502146. Epub 2019 Aug 16. Nephron. 2019. PMID: 31422399 Review.
Cited by
-
Convert your favorite protein modeling program into a mutation predictor: "MODICT".BMC Bioinformatics. 2016 Oct 19;17(1):425. doi: 10.1186/s12859-016-1286-0. BMC Bioinformatics. 2016. PMID: 27760515 Free PMC article.
-
Cystic Kidney Diseases That Require a Differential Diagnosis from Autosomal Dominant Polycystic Kidney Disease (ADPKD).J Clin Med. 2022 Nov 3;11(21):6528. doi: 10.3390/jcm11216528. J Clin Med. 2022. PMID: 36362756 Free PMC article. Review.
-
Autosomal Dominant Tubulointerstitial Kidney Disease.Adv Chronic Kidney Dis. 2017 Mar;24(2):86-93. doi: 10.1053/j.ackd.2016.11.012. Adv Chronic Kidney Dis. 2017. PMID: 28284384 Free PMC article. Review.
-
Autosomal dominant tubulointerstitial kidney disease: more than just HNF1β.Pediatr Nephrol. 2022 May;37(5):933-946. doi: 10.1007/s00467-021-05118-4. Epub 2021 May 22. Pediatr Nephrol. 2022. PMID: 34021396 Free PMC article. Review.
-
Autosomal dominant mutation in the signal peptide of renin in a kindred with anemia, hyperuricemia, and CKD.Am J Kidney Dis. 2011 Nov;58(5):821-5. doi: 10.1053/j.ajkd.2011.06.029. Epub 2011 Sep 8. Am J Kidney Dis. 2011. PMID: 21903317 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

