Neuronal migration disorders in microcephalic osteodysplastic primordial dwarfism type I/III
- PMID: 20857301
- PMCID: PMC3059390
- DOI: 10.1007/s00401-010-0748-0
Neuronal migration disorders in microcephalic osteodysplastic primordial dwarfism type I/III
Abstract
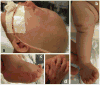
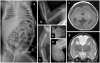
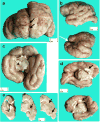
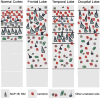
Microcephalic osteodysplastic primordial dwarfism (MOPD) is a rare microlissencephaly syndrome, with at least two distinct phenotypic and genetic types. MOPD type II is caused by pericentrin mutations, while types I and III appear to represent a distinct entity (MOPD I/III) with variably penetrant phenotypes and unknown genetic basis. The neuropathology of MOPD I/III is little understood, especially in comparison to other forms of lissencephaly. Here, we report postmortem brain findings in an 11-month-old female infant with MOPD I/III. The cerebral cortex was diffusely pachygyric, with a right parietal porencephalic lesion. Histologically, the cortex was abnormally thick and disorganized. Distinct malformations were observed in different cerebral lobes, as characterized using layer-specific neuronal markers. Frontal cortex was severely disorganized and coated with extensive leptomeningeal glioneuronal heterotopia. Temporal cortex had a relatively normal 6-layered pattern, despite cortical thickening. Occipital cortex was variably affected. The corpus callosum was extremely hypoplastic. Brainstem and cerebellar malformations were also present, as well as old necrotic foci. Findings in this case suggest that the cortical malformation in MOPD I/III is distinct from other forms of pachygyria-lissencephaly.
Figures




Similar articles
-
Microcephalic osteodysplastic primordial dwarfism type II and pachygyria: Morphometric analysis in a 2-year-old girl.Am J Med Genet A. 2020 Oct;182(10):2372-2376. doi: 10.1002/ajmg.a.61771. Epub 2020 Aug 3. Am J Med Genet A. 2020. PMID: 32744776
-
Microcephalic osteodysplastic primordial dwarfism type I/III in sibs.J Med Genet. 1991 Nov;28(11):795-800. doi: 10.1136/jmg.28.11.795. J Med Genet. 1991. PMID: 1770539 Free PMC article.
-
Microcephalic osteodysplastic primordial dwarfism type II: Additional nine patients with implications on phenotype and genotype correlation.Am J Med Genet A. 2020 Jun;182(6):1407-1420. doi: 10.1002/ajmg.a.61585. Epub 2020 Apr 8. Am J Med Genet A. 2020. PMID: 32267100
-
Microcephalic osteodysplastic primordial dwarfism Taybi-Linder type: report of four cases and review of the literature.Am J Med Genet. 1998 Oct 30;80(1):16-24. Am J Med Genet. 1998. PMID: 9800907 Review.
-
Microcephalic osteodysplastic primordial dwarfism type II: report of three cases and review.Am J Med Genet. 1998 Oct 30;80(1):25-31. doi: 10.1002/(sici)1096-8628(19981102)80:1<25::aid-ajmg5>3.0.co;2-0. Am J Med Genet. 1998. PMID: 9800908 Review.
Cited by
-
Research models of neurodevelopmental disorders: The right model in the right place.Front Neurosci. 2022 Oct 20;16:1031075. doi: 10.3389/fnins.2022.1031075. eCollection 2022. Front Neurosci. 2022. PMID: 36340790 Free PMC article. Review.
-
Posterior fossa in primary microcephaly: relationships between forebrain and mid-hindbrain size in 110 patients.Neuropediatrics. 2014 Apr;45(2):93-101. doi: 10.1055/s-0033-1360483. Epub 2013 Nov 14. Neuropediatrics. 2014. PMID: 24234199 Free PMC article.
-
Malformations of Cerebral Cortex Development: Molecules and Mechanisms.Annu Rev Pathol. 2019 Jan 24;14:293-318. doi: 10.1146/annurev-pathmechdis-012418-012927. Annu Rev Pathol. 2019. PMID: 30677308 Free PMC article. Review.
-
Cellular and axonal diversity in molecular layer heterotopia of the rat cerebellar vermis.Biomed Res Int. 2013;2013:805467. doi: 10.1155/2013/805467. Epub 2013 Sep 26. Biomed Res Int. 2013. PMID: 24191251 Free PMC article.
-
Microcephalic osteodysplastic primordial dwarfism type I with biallelic mutations in the RNU4ATAC gene.Clin Genet. 2012 Aug;82(2):140-6. doi: 10.1111/j.1399-0004.2011.01756.x. Epub 2011 Aug 28. Clin Genet. 2012. PMID: 21815888 Free PMC article.
References
-
- Anderson SA, Kaznowski CE, Horn C, et al. Distinct origins of neocortical projection neurons and interneurons in vivo. Cereb Cortex. 2002;12:702–709. - PubMed
-
- Barkovich AJ, Koch TK, Carrol CL. The spectrum of lissencephaly: report of ten patients analyzed by magnetic resonance imaging. Ann Neurol. 1991;30:139–146. - PubMed
-
- Dobyns WB, Stratton RF, Greenberg R. Syndromes with lissencephaly I: Miller–Dieker and Norman–Roberts syndromes and isolated lissencephaly. Am J Med Genet. 1984;18:509–526. - PubMed
-
- Donmez FY, Yildirim M, Erkek N. Hippocampal abnormalities associated with various congenital malformations. Childs Nerv Syst. 2009;25:933–999. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources

