Unbalanced der(5)t(5;20) translocation associated with megalencephaly, perisylvian polymicrogyria, polydactyly and hydrocephalus
- PMID: 20503325
- PMCID: PMC2908594
- DOI: 10.1002/ajmg.a.33408
Unbalanced der(5)t(5;20) translocation associated with megalencephaly, perisylvian polymicrogyria, polydactyly and hydrocephalus
Abstract
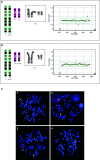
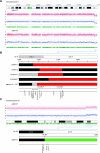
The combination of megalencephaly, perisylvian polymicrogyria, polydactyly and hydrocephalus (MPPH) is a rare syndrome of unknown cause. We observed two first cousins affected by an MPPH-like phenotype with a submicroscopic chromosome 5q35 deletion as a result of an unbalanced der(5)t(5;20)(q35.2;q13.3) translocation, including the NSD1 Sotos syndrome locus. We describe the phenotype and the deletion breakpoints of the two MPPH-like patients and compare these with five unrelated MPPH and Sotos patients harboring a 5q35 microdeletion. Mapping of the breakpoints in the two cousins was performed by MLPA, FISH, high density SNP-arrays and Q-PCR for the 5q35 deletion and 20q13 duplication. The 5q35 deletion area of the two cousins almost completely overlaps with earlier described patients with an atypical Sotos microdeletion, except for the DRD1 gene. The five unrelated MPPH patients neither showed submicroscopic chromosomal aberrations nor DRD1 mutations. We reviewed the brain MRI of 10 Sotos patients and did not detect polymicrogyria in any of them. In our two cousins, the MPPH-like phenotype is probably caused by the contribution of genes on both chromosome 5q35 and 20q13. Some patients with MPPH may harbor a submicroscopic chromosomal aberration and therefore high-resolution array analysis should be part of the diagnostic workup.
(c) 2010 Wiley-Liss, Inc.
Figures

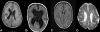
Similar articles
-
Significant overlap and possible identity of macrocephaly capillary malformation and megalencephaly polymicrogyria-polydactyly hydrocephalus syndromes.Am J Med Genet A. 2009 May;149A(5):868-76. doi: 10.1002/ajmg.a.32732. Am J Med Genet A. 2009. PMID: 19353582
-
Maternal mosaicism underlies the inheritance of a rare germline AKT3 variant which is responsible for megalencephaly-polymicrogyria-polydactyly-hydrocephalus syndrome in two Roma half-siblings.Exp Mol Pathol. 2020 Aug;115:104471. doi: 10.1016/j.yexmp.2020.104471. Epub 2020 May 21. Exp Mol Pathol. 2020. PMID: 32446860
-
Prenatal Ultrasound Diagnosis of Megalencephaly-Polymicrogyria-Polydactyly-Hydrocephalus Syndrome with Persistent Hyperplastic Primary Vitreous: A Case Report.Fetal Diagn Ther. 2024;51(2):154-158. doi: 10.1159/000535509. Epub 2023 Nov 26. Fetal Diagn Ther. 2024. PMID: 38008077
-
Proximal variants in CCND2 associated with microcephaly, short stature, and developmental delay: A case series and review of inverse brain growth phenotypes.Am J Med Genet A. 2021 Sep;185(9):2719-2738. doi: 10.1002/ajmg.a.62362. Epub 2021 Jun 4. Am J Med Genet A. 2021. PMID: 34087052 Free PMC article. Review.
-
Chromosome 5q subtelomeric deletion syndrome.Am J Med Genet C Semin Med Genet. 2007 Nov 15;145C(4):372-6. doi: 10.1002/ajmg.c.30151. Am J Med Genet C Semin Med Genet. 2007. PMID: 17910075 Review.
Cited by
-
Four-year follow-up of megalencephaly, polymicrogyria, postaxial polydactyly and hydrocephalus (MPPH) syndrome.BMJ Case Rep. 2013 Oct 3;2013:bcr2012007826. doi: 10.1136/bcr-2012-007826. BMJ Case Rep. 2013. PMID: 24092603 Free PMC article.
-
The genetic basis of hydrocephalus: genes, pathways, mechanisms, and global impact.Fluids Barriers CNS. 2024 Mar 4;21(1):24. doi: 10.1186/s12987-024-00513-z. Fluids Barriers CNS. 2024. PMID: 38439105 Free PMC article. Review.
-
Sotos Syndrome: Deep Neuroimaging Phenotyping Reveals a High Prevalence of Malformations of Cortical Development.AJNR Am J Neuroradiol. 2024 Oct 3;45(10):1570-1577. doi: 10.3174/ajnr.A8364. AJNR Am J Neuroradiol. 2024. PMID: 39147584
References
-
- Addor MC, Castagne C, Micheli JL, Schorderet DF. Partial trisomy 20q in a newborn with dextrocardia. Genet Couns. 2002;13:433–440. - PubMed
-
- Barkovich AJ, Kuzniecky RI, Jackson GD, Guerrini R, Dobyns WB. A developmental and genetic classification for malformations of cortical development. Neurology. 2005;65:1873–1887. - PubMed
-
- Barth PG. International review of Child neurology series (ICNA) Mac Keith Press; London: 2003. Disorders of Neuronal Migration.
-
- Bastepe M. The GNAS locus and pseudohypoparathyroidism. Adv Exp Med Biol. 2008;626:27–40. - PubMed
-
- Bonthron DT, Hayward BE. Characterization of TH1 and CTSZ, two non-imprinted genes downstream of GNAS1 in chromosome 20q13. Hum Genet. 2000;107:165–175. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

