Genetic and ultrastructural studies in dilated cardiomyopathy patients: a large deletion in the lamin A/C gene is associated with cardiomyocyte nuclear envelope disruption
- PMID: 20127487
- PMCID: PMC3934843
- DOI: 10.1007/s00395-010-0085-4
Genetic and ultrastructural studies in dilated cardiomyopathy patients: a large deletion in the lamin A/C gene is associated with cardiomyocyte nuclear envelope disruption
Abstract
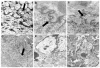
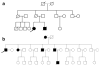
Major nuclear envelope abnormalities, such as disruption and/or presence of intranuclear organelles, have rarely been described in cardiomyocytes from dilated cardiomyopathy (DCM) patients. In this study, we screened a series of 25 unrelated DCM patient samples for (a) cardiomyocyte nuclear abnormalities and (b) mutations in LMNA and TMPO as they are two DCM-causing genes that encode proteins involved in maintaining nuclear envelope architecture. Among the 25 heart samples investigated, we identified major cardiomyocyte nuclear abnormalities in 8 patients. Direct sequencing allowed the detection of three heterozygous LMNA mutations (p.D192G, p.Q353K and p.R541S) in three patients. By multiplex ligation-dependant probe amplification (MLPA)/quantitative real-time PCR, we found a heterozygous deletion encompassing exons 3-12 of the LMNA gene in one patient. Immunostaining demonstrated that this deletion led to a decrease in lamin A/C expression in cardiomyocytes from this patient. This LMNA deletion as well as the p.D192G mutation was found in patients displaying major cardiomyocyte nuclear envelope abnormalities, while the p.Q353K and p.R541S mutations were found in patients without specific nuclear envelope abnormalities. None of the DCM patients included in the study carried a mutation in the TMPO gene. Taken together, we found no evidence of a genotype-phenotype relationship between the onset and the severity of DCM, the presence of nuclear abnormalities and the presence or absence of LMNA mutations. We demonstrated that a large deletion in LMNA associated with reduced levels of the protein in the nuclear envelope suggesting a haploinsufficiency mechanism can lead to cardiomyocyte nuclear envelope disruption and thus underlie the pathogenesis of DCM.
Conflict of interest statement
Figures

Similar articles
-
In vivo and in vitro examination of the functional significances of novel lamin gene mutations in heart failure patients.J Med Genet. 2005 Aug;42(8):639-47. doi: 10.1136/jmg.2004.023283. J Med Genet. 2005. PMID: 16061563 Free PMC article.
-
Clinical and functional characterization of a novel mutation in lamin a/c gene in a multigenerational family with arrhythmogenic cardiac laminopathy.PLoS One. 2015 Apr 2;10(4):e0121723. doi: 10.1371/journal.pone.0121723. eCollection 2015. PLoS One. 2015. PMID: 25837155 Free PMC article.
-
Obliteration of cardiomyocyte nuclear architecture in a patient with LMNA gene mutation.J Neurol Sci. 2008 Aug 15;271(1-2):91-6. doi: 10.1016/j.jns.2008.03.017. Epub 2008 May 27. J Neurol Sci. 2008. PMID: 18502446
-
Lamin A/C mutations in dilated cardiomyopathy.Cardiol J. 2014;21(4):331-42. doi: 10.5603/CJ.a2014.0037. Epub 2014 May 20. Cardiol J. 2014. PMID: 24846508 Review.
-
Lamin and the heart.Heart. 2018 Mar;104(6):468-479. doi: 10.1136/heartjnl-2017-312338. Epub 2017 Nov 25. Heart. 2018. PMID: 29175975 Review.
Cited by
-
Lamin A mutation impairs interaction with nucleoporin NUP155 and disrupts nucleocytoplasmic transport in atrial fibrillation.Hum Mutat. 2019 Mar;40(3):310-325. doi: 10.1002/humu.23691. Epub 2018 Dec 8. Hum Mutat. 2019. PMID: 30488537 Free PMC article.
-
Nuclear damage in LMNA mutant iPSC-derived cardiomyocytes is associated with impaired lamin localization to the nuclear envelope.Mol Biol Cell. 2023 Aug 16;34(12):mbcE21100527. doi: 10.1091/mbc.E21-10-0527. Online ahead of print. Mol Biol Cell. 2023. PMID: 37585285 Free PMC article.
-
Implications and Assessment of the Elastic Behavior of Lamins in Laminopathies.Cells. 2016 Oct 14;5(4):37. doi: 10.3390/cells5040037. Cells. 2016. PMID: 27754432 Free PMC article. Review.
-
Ablation of SUN2-containing LINC complexes drives cardiac hypertrophy without interstitial fibrosis.Mol Biol Cell. 2019 Jul 1;30(14):1664-1675. doi: 10.1091/mbc.E18-07-0438. Epub 2019 May 15. Mol Biol Cell. 2019. PMID: 31091167 Free PMC article.
-
EH-myomesin splice isoform is a novel marker for dilated cardiomyopathy.Basic Res Cardiol. 2011 Mar;106(2):233-47. doi: 10.1007/s00395-010-0131-2. Epub 2010 Nov 11. Basic Res Cardiol. 2011. PMID: 21069531 Free PMC article.
References
-
- Arbustini E, Gavazzi A, Pozzi R, Grasso M, Pucci A, Campana C, Graziano G, Martinetti M, Cuccia M, Salvaneschi L, Martinelli L, Montemartini C, Vigano M. The morphologic spectrum of dilated cardiomyopathy and its relation to immune-response genes. Am J Cardiol. 1989;64:991–995. - PubMed
-
- Arbustini E, Pilotto A, Repetto A, Grasso M, Negri A, Diegoli M, Campana C, Scelsi L, Baldini E, Gavazzi A, Tavazzi L. Autosomal dominant dilated cardiomyopathy with atrioventricular block: a lamin A/C defect-related disease. J Am Coll Cardiol. 2002;39:981–990. - PubMed
-
- Arimura T, Hayashi T, Matsumoto Y, Shibata H, Hiroi S, Nakamura T, Inagaki N, Hinohara K, Takahashi M, Manatsu S, Sasaoka T, Izumi T, Bonne G, Schwartz K, Kimura A. Structural analysis of four and half LIM protein-2 in dilated cardiomyopathy. Biochem Biophys Res Commun. 2007;357:162–167. - PubMed
-
- Baandrup U, Florio RA, Roters F, Olsen EG. Electron microscopic investigation of endomyocardial biopsy samples in hypertrophy and cardiomyopathy. A semiquantitative study in 48 patients. Circulation. 1981;63:1289–1298. - PubMed
-
- Bécane HM, Bonne G, Varnous S, Muchir A, Ortega V, Hammouda EH, Urtizberea JA, Lavergne T, Fardeau M, Eymard B, Weber S, Schwartz K, Duboc D. High incidence of sudden death with conduction system and myocardial disease due to lamins A and C gene mutation. J Pac Clin Electrophysiol. 2000;23:1661–1666. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

