Computerized counseling for folate knowledge and use: a randomized controlled trial
- PMID: 19000845
- PMCID: PMC2593421
- DOI: 10.1016/j.amepre.2008.06.034
Computerized counseling for folate knowledge and use: a randomized controlled trial
Abstract
Background: Periconception folate supplementation significantly reduces the risk of neural-tube defects, but few U.S. women start folate supplementation before pregnancy, and the amount of clinician time available to counsel patients about folate is limited. This study evaluated whether computer-assisted counseling and the provision of free folate tablets increases women's knowledge and use of folate supplements.
Design: Randomized controlled trial; follow-up began 6 months after enrollment and was completed on average 7 months after enrollment.
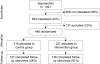
Setting/participants: A total of 446 women, aged 18-45 years, were recruited from two urgent care clinics in San Francisco from March to July 2005 (data collection was completed in 2006; data were analyzed in 2007).
Intervention: Participants received a 15-minute computerized educational session and 200 folate tablets.
Main outcome measures: The primary outcome was the knowledge that folate can prevent birth defects; secondary outcomes included the self-reported use of a folate supplement at follow-up.
Results: At follow-up, women in the intervention group were more likely to know that folate prevents birth defects (46% vs 27%, relative risk [RR]=1.72, 95% CI=1.32, 2.23); to know that folate is most important in early pregnancy (36% vs 17%, RR=2.11, 95% CI=1.50, 2.97); and to report the recent use of a folate supplement (32% vs 21%, RR=1.54, 95% CI=1.12, 2.13).
Conclusions: A one-time, brief, computerized counseling session about folate with the provision of free folate tablets increased the knowledge and use of folate supplements among women > or =6 months later.
Trial registration: NCT00177515.
Comment in
-
Elimination of folic acid-preventable neural tube defects.Am J Prev Med. 2008 Dec;35(6):606-7. doi: 10.1016/j.amepre.2008.09.017. Am J Prev Med. 2008. PMID: 19000851 No abstract available.
Similar articles
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Pre-conception Folic Acid and Multivitamin Supplementation for the Primary and Secondary Prevention of Neural Tube Defects and Other Folic Acid-Sensitive Congenital Anomalies.J Obstet Gynaecol Can. 2015 Jun;37(6):534-52. doi: 10.1016/s1701-2163(15)30230-9. J Obstet Gynaecol Can. 2015. PMID: 26334606 English, French.
-
The use of folic acid and other vitamins before and during pregnancy in a group of women in Melbourne, Australia.Midwifery. 2009 Apr;25(2):134-46. doi: 10.1016/j.midw.2007.01.019. Epub 2007 May 31. Midwifery. 2009. PMID: 17543431
-
WITHDRAWN: Periconceptional supplementation with folate and/or multivitamins for preventing neural tube defects.Cochrane Database Syst Rev. 2011 Apr 13;2011(4):CD001056. doi: 10.1002/14651858.CD001056.pub2. Cochrane Database Syst Rev. 2011. PMID: 21491380 Free PMC article. Review.
-
Periconceptional supplementation with folate and/or multivitamins for preventing neural tube defects.Cochrane Database Syst Rev. 2001;(3):CD001056. doi: 10.1002/14651858.CD001056. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2011 Apr 13;(4):CD001056. doi: 10.1002/14651858.CD001056.pub2. PMID: 11686974 Updated. Review.
Cited by
-
Effects and safety of periconceptional folate supplementation for preventing birth defects.Cochrane Database Syst Rev. 2010 Oct 6;(10):CD007950. doi: 10.1002/14651858.CD007950.pub2. Cochrane Database Syst Rev. 2010. Update in: Cochrane Database Syst Rev. 2015 Dec 14;(12):CD007950. doi: 10.1002/14651858.CD007950.pub3. PMID: 20927767 Free PMC article. Updated. Review.
-
Acceptability and feasibility of a planned preconception weight loss intervention in women with long-acting reversible contraception: the Plan-it mixed-methods study.Health Technol Assess. 2023 Jan;27(1):1-224. doi: 10.3310/NKIX8285. Health Technol Assess. 2023. PMID: 36688498 Free PMC article. Clinical Trial.
-
Impact of a pharmacist-directed educational program on the long-term knowledge and use of folic acid among college women: a 12-month follow-up study.Pharm Pract (Granada). 2012 Apr;10(2):105-9. doi: 10.4321/s1886-36552012000200007. Epub 2012 Jun 30. Pharm Pract (Granada). 2012. PMID: 24155825 Free PMC article.
-
Decreased incidence of myelomeningocele at birth: effect of folic acid recommendations or prenatal diagnostics?Childs Nerv Syst. 2011 Nov;27(11):1951-5. doi: 10.1007/s00381-011-1473-2. Epub 2011 May 7. Childs Nerv Syst. 2011. PMID: 21552997
-
Women's perspectives on counseling about risks for medication-induced birth defects.Birth Defects Res A Clin Mol Teratol. 2010 Jan;88(1):64-9. doi: 10.1002/bdra.20618. Birth Defects Res A Clin Mol Teratol. 2010. PMID: 19637252 Free PMC article.
References
-
- CDC Use of dietary supplements containing folic acid among women of childbearing age—U.S., 2005. MMWR Morb Mortal Wkly Rep. 2005;54(38):955–8. - PubMed
-
- Lumley J, Watson L, Watson M, Bower C. Periconceptional supplementation with folate and/or multivitamins for preventing neural tube defects. Cochrane Database Syst Rev. 2001;(3) CD001056. - PubMed
-
- Finer LB, Henshaw SK. Disparities in rates of unintended pregnancy in the U.S., 1994 and 2001. Perspect Sex Reprod Health. 2006;38(2):90–6. - PubMed
-
- CDC Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects. MMWR Morb Mortal Wkly Rep. 1992;41(RR14):1–7. - PubMed
-
- Chacko MR, Anding R, Kozinetz CA, Grover JL, Smith PB. Neural tube defects: knowledge and preconceptional prevention practices in minority young women. Pediatrics. 2003;112(3 Pt 1):536–42. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical