Combinatorial antileukemic disruption of oxidative homeostasis and mitochondrial stability by the redox reactive thalidomide 2-(2,4-difluoro-phenyl)-4,5,6,7-tetrafluoro-1H-isoindole-1,3(2H)-dione (CPS49) and flavopiridol
- PMID: 18556456
- PMCID: PMC2778846
- DOI: 10.1124/mol.107.040808
Combinatorial antileukemic disruption of oxidative homeostasis and mitochondrial stability by the redox reactive thalidomide 2-(2,4-difluoro-phenyl)-4,5,6,7-tetrafluoro-1H-isoindole-1,3(2H)-dione (CPS49) and flavopiridol
Abstract
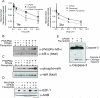
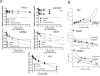
2-(2,4-Difluoro-phenyl)-4,5,6,7-tetrafluoro-1H-isoindole-1,3(2H)-dione (CPS49) is a member of a recently identified class of redox-reactive thalidomide analogs that show selective killing of leukemic cells by increasing intracellular reactive oxygen species (ROS) and targeting multiple transcriptional pathways. Flavopiridol is a semisynthetic flavonoid that inhibits cyclin-dependent kinases and also shows selective lethality against leukemic cells. The purpose of this study is to explore the efficacy and mechanism of action of the combinatorial use of the redox-reactive thalidomide CPS49 and the cyclin-dependent kinase inhibitor flavopiridol as a selective antileukemic therapeutic strategy. In combination, CPS49 and flavopiridol were found to induce selective cytotoxicity associated with mitochondrial dysfunction and elevations of ROS in leukemic cells ranging from additive to synergistic activity at low micromolar concentrations. Highest synergy was observed at the level of ROS generation with a strong correlation between cell-specific cytotoxicity and reciprocal coupling of drug-induced ROS elevation with glutathione depletion. Examination of the transcriptional targeting of CPS49 and flavopiridol combinations reveals that the drugs act in concert to initiate a cell specific transcriptional program that manipulates nuclear factor-kappaB (NF-kappaB), E2F-1, and p73 activity to promote enhanced mitochondrial instability by simultaneously elevating the expression of the proapoptotic factors BAX, BAD, p73, and PUMA while depressing expression of the antiapoptotic genes MCL1, XIAP, BCL-xL, SURVIVIN, and MDM2. The coadministration of CPS49 and flavopiridol acts through coordinate targeting of transcriptional pathways that enforce selective mitochondrial dysfunction and ROS elevation and is therefore a promising new therapeutic combination that warrants further preclinical exploration.
Figures



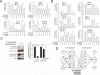
Similar articles
-
Low doses of CPS49 and flavopiridol combination as potential treatment for advanced prostate cancer.Curr Pharm Biotechnol. 2015;16(6):553-63. doi: 10.2174/138920101606150407114407. Curr Pharm Biotechnol. 2015. Retraction in: Curr Pharm Biotechnol. 2019;20(12):1072. doi: 10.2174/138920102012190919103433. PMID: 25860066 Retracted.
-
Contribution of disruption of the nuclear factor-kappaB pathway to induction of apoptosis in human leukemia cells by histone deacetylase inhibitors and flavopiridol.Mol Pharmacol. 2004 Oct;66(4):956-63. doi: 10.1124/mol.104.002014. Epub 2004 Jul 2. Mol Pharmacol. 2004. PMID: 15235103
-
The small-molecule Bcl-2 inhibitor HA14-1 interacts synergistically with flavopiridol to induce mitochondrial injury and apoptosis in human myeloma cells through a free radical-dependent and Jun NH2-terminal kinase-dependent mechanism.Mol Cancer Ther. 2004 Dec;3(12):1513-24. Mol Cancer Ther. 2004. PMID: 15634644
-
Mechanisms of action of flavopiridol.Crit Rev Oncol Hematol. 2001 May;38(2):139-70. doi: 10.1016/s1040-8428(00)00124-4. Crit Rev Oncol Hematol. 2001. PMID: 11311660 Review.
-
Flavopiridol: pleiotropic biological effects enhance its anti-cancer activity.Anticancer Drugs. 2004 Jun;15(5):411-9. doi: 10.1097/01.cad.0000127332.06439.47. Anticancer Drugs. 2004. PMID: 15166614 Review.
Cited by
-
Increasing the activity of copper(II) complexes against Leishmania through lipophilicity and pro-oxidant ability.J Biol Inorg Chem. 2012 Jan;17(1):107-12. doi: 10.1007/s00775-011-0834-3. Epub 2011 Aug 25. J Biol Inorg Chem. 2012. PMID: 21866394
-
Mechanism of immunomodulatory drugs' action in the treatment of multiple myeloma.Acta Biochim Biophys Sin (Shanghai). 2014 Mar;46(3):240-53. doi: 10.1093/abbs/gmt142. Epub 2013 Dec 29. Acta Biochim Biophys Sin (Shanghai). 2014. PMID: 24374776 Free PMC article. Review.
-
Cancer drug resistance: redox resetting renders a way.Oncotarget. 2016 Jul 5;7(27):42740-42761. doi: 10.18632/oncotarget.8600. Oncotarget. 2016. PMID: 27057637 Free PMC article. Review.
-
Preformulation study of NSC-726796.AAPS PharmSciTech. 2012 Jun;13(2):661-73. doi: 10.1208/s12249-012-9784-5. Epub 2012 May 3. AAPS PharmSciTech. 2012. PMID: 22552929 Free PMC article.
References
-
- Blagosklonny MV. Flavopiridol, an Inhibitor of Transcription: Implications, Problems and Solutions. Cell Cycle. 2004;3:1537–1542. - PubMed
-
- Chao SH, Price DH. Flavopiridol Inactivates P-TEFb and Blocks Most RNA Polymerase II Transcription in Vivo. J Biol Chem. 2001;276:31793–31799. - PubMed
-
- Chou TC, Talalay P. Quantitative Analysis of Dose-Effect Relationships: the Combined Effects of Multiple Drugs or Enzyme Inhibitors. Adv Enzyme Regul. 1984;22:27–55. - PubMed
-
- Craig RW. MCL1 Provides a Window on the Role of the BCL2 Family in Cell Proliferation, Differentiation and Tumorigenesis. Leukemia. 2002;16:444–454. - PubMed
-
- Dai Y, Grant S. Cyclin-Dependent Kinase Inhibitors. Curr Opin Pharmacol. 2003;3:362–370. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

