Regulation of neurite outgrowth by G(i/o) signaling pathways
- PMID: 18508528
- PMCID: PMC3068557
- DOI: 10.2741/3022
Regulation of neurite outgrowth by G(i/o) signaling pathways
Abstract
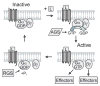
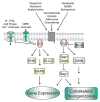
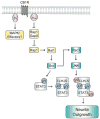
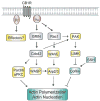
Neurogenesis is a long and winding journey. A neural progenitor cell migrates long distances, differentiates by forming a single axon and multiple dendrites, undergoes maturation, and ultimately survives. The initial formation of neurites during neuronal differentiation, commonly referred to as "neurite outgrowth," can be induced by a large repertoire of signals that stimulate an array of receptors and downstream signaling pathways. The G(i/o) family of heterotrimeric G-proteins are abundantly expressed in the brain and enriched at neuronal growth cones. Recent evidence has uncovered several G(i/o)-coupled receptors that induce neurite outgrowth and has begun to elucidate the underlying molecular mechanisms. Emerging data suggests that signals from several G(i/o)-coupled receptors converge at the transcription factor STAT3 to regulate neurite outgrowth and at Rac1 and Cdc42 to regulate cytoskeletal reorganization. Physiologically, signaling through G(i/o)-coupled cannabinoid receptors is critical for pro percentral nervous system development. As the mechanisms by which G(i/o)-coupled receptors regulate neurite outgrowth are clarified, it is becoming evident that modulating signals from G(i/o) and their receptors has great potential for the treatment of neurodegenerative diseases.
Figures
Similar articles
-
The G alpha(o/i)-coupled cannabinoid receptor-mediated neurite outgrowth involves Rap regulation of Src and Stat3.J Biol Chem. 2005 Sep 30;280(39):33426-34. doi: 10.1074/jbc.M502812200. Epub 2005 Jul 26. J Biol Chem. 2005. PMID: 16046413
-
Role of the Go/i signaling network in the regulation of neurite outgrowth.Can J Physiol Pharmacol. 2006 Jul;84(7):687-94. doi: 10.1139/y06-025. Can J Physiol Pharmacol. 2006. PMID: 16998532 Review.
-
Cannabinoid receptor-induced neurite outgrowth is mediated by Rap1 activation through G(alpha)o/i-triggered proteasomal degradation of Rap1GAPII.J Biol Chem. 2005 Mar 25;280(12):11413-21. doi: 10.1074/jbc.M411521200. Epub 2005 Jan 18. J Biol Chem. 2005. PMID: 15657046
-
δ-opioid receptor activation leads to neurite outgrowth and neuronal differentiation via a STAT5B-Gαi/o pathway.J Neurochem. 2013 Nov;127(3):329-41. doi: 10.1111/jnc.12386. Epub 2013 Sep 18. J Neurochem. 2013. PMID: 23906478
-
Neuro2A differentiation by Galphai/o pathway.Sci Signal. 2009 Jan 20;2(54):cm1. doi: 10.1126/scisignal.254cm1. Sci Signal. 2009. PMID: 19155528 Free PMC article. Review.
Cited by
-
Pediatric Encephalopathy: Clinical, Biochemical and Cellular Insights into the Role of Gln52 of GNAO1 and GNAI1 for the Dominant Disease.Cells. 2021 Oct 14;10(10):2749. doi: 10.3390/cells10102749. Cells. 2021. PMID: 34685729 Free PMC article.
-
Genes encoding heterotrimeric G-proteins are associated with gray matter volume variations in the medial frontal cortex.Cereb Cortex. 2013 May;23(5):1025-30. doi: 10.1093/cercor/bhs061. Epub 2012 Apr 17. Cereb Cortex. 2013. PMID: 22510535 Free PMC article.
-
Subcellular optogenetics - controlling signaling and single-cell behavior.J Cell Sci. 2015 Jan 1;128(1):15-25. doi: 10.1242/jcs.154435. Epub 2014 Nov 28. J Cell Sci. 2015. PMID: 25433038 Free PMC article. Review.
-
Can case study approaches speed implementation of the NRC report: "Toxicity Testing in the 21st Century: A Vision and a Strategy?".ALTEX. 2011;28(3):175-82. doi: 10.14573/altex.2011.3.175. ALTEX. 2011. PMID: 21993955 Free PMC article.
-
Diacylglycerol lipase-alpha and -beta control neurite outgrowth in neuro-2a cells through distinct molecular mechanisms.Mol Pharmacol. 2011 Jul;80(1):60-7. doi: 10.1124/mol.110.070458. Epub 2011 Apr 14. Mol Pharmacol. 2011. PMID: 21493725 Free PMC article.
References
-
- Govek EE, Newey SE, Van Aelst L. The role of the Rho GTPases in neuronal development. Genes Dev. 2005;19(1):1–49. - PubMed
-
- Ohnuma S, Harris WA. Neurogenesis and the cell cycle. Neuron. 2003;40(2):199–208. - PubMed
-
- Diez del Corral R, Storey KG. Markers in vertebrate neurogenesis. Nat Rev Neurosci. 2001;2(11):835–9. - PubMed
-
- da Silva JS, Dotti CG. Breaking the neuronal sphere: regulation of the actin cytoskeleton in neuritogenesis. Nat Rev Neurosci. 2002;3(9):694–704. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
