Pontocerebellar hypoplasia type 2: a neuropathological update
- PMID: 17641900
- PMCID: PMC2039791
- DOI: 10.1007/s00401-007-0263-0
Pontocerebellar hypoplasia type 2: a neuropathological update
Abstract
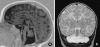
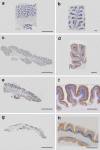
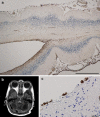
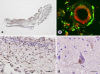
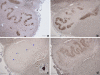
Pontocerebellar hypoplasia type 2 (PCH-2; MIM 277470), an autosomal recessive neurodegeneration with fetal onset, was studied in six autopsies with ages at death ranging between 1 and 22 years. Three patients were distantly related. A case of olivopontocerebellar hypoplasia (OPCH; MIM 225753) was studied for comparison. Typical findings are: short cerebellar folia with poor branching ("hypoplasia"), relative sparing of the vermis, sharply demarcated areas of full thickness loss of cerebellar cortex probably resulting from regression at an early stage of development, segmental loss of dentate nuclei with preserved islands and reactive changes, segmental loss in the inferior olivary nucleus with reactive changes, loss of ventral pontine nuclei with near absence of transverse pontine fibers and sparing of spinal anterior horn cells. Variable findings are: cystic cerebellar degeneration, found in two, with vascular changes limited to the cerebellum in one. Comparison to olivopontocerebellar hypoplasia (OPCH) strongly suggests a continuum of pathology between this disorder and PCH-2. Immunohistochemical evaluation of the endoplasmic reticulum stress response is negative. We conclude that the neuropathological findings in PCH-2 are sufficiently specific to enable an unequivocal diagnosis based on neuropathology.
Figures






Comment in
-
Progress on pontocerebellar hypoplasia.Acta Neuropathol. 2007 Oct;114(4):401-2. doi: 10.1007/s00401-007-0282-x. Epub 2007 Aug 21. Acta Neuropathol. 2007. PMID: 17710422 Review. No abstract available.
Similar articles
-
Severe, fetal-onset form of olivopontocerebellar hypoplasia in three sibs: PCH type 5?Am J Med Genet A. 2006 Mar 15;140(6):594-603. doi: 10.1002/ajmg.a.31095. Am J Med Genet A. 2006. PMID: 16470708
-
Pontocerebellar hypoplasia type 3 with tetralogy of Fallot.Brain Dev. 2012 May;34(5):392-5. doi: 10.1016/j.braindev.2011.07.011. Epub 2011 Aug 30. Brain Dev. 2012. PMID: 21880448
-
Autopsy case of later-onset pontocerebellar hypoplasia type 1: pontine atrophy and pyramidal tract involvement.J Child Neurol. 2010 Nov;25(11):1429-34. doi: 10.1177/0883073810372991. Epub 2010 Jun 17. J Child Neurol. 2010. PMID: 20558670
-
Pontocerebellar hypoplasias. An overview of a group of inherited neurodegenerative disorders with fetal onset.Brain Dev. 1993 Nov-Dec;15(6):411-22. doi: 10.1016/0387-7604(93)90080-r. Brain Dev. 1993. PMID: 8147499 Review.
-
Pontocerebellar hypoplasia: review of classification and genetics, and exclusion of several genes known to be important for cerebellar development.J Child Neurol. 2011 Mar;26(3):288-94. doi: 10.1177/0883073810380047. J Child Neurol. 2011. PMID: 21383226 Review.
Cited by
-
Clinical, neuroradiological and genetic findings in pontocerebellar hypoplasia.Brain. 2011 Jan;134(Pt 1):143-56. doi: 10.1093/brain/awq287. Epub 2010 Oct 15. Brain. 2011. PMID: 20952379 Free PMC article.
-
Pathogenic variants in AIMP1 cause pontocerebellar hypoplasia.Neurogenetics. 2019 May;20(2):103-108. doi: 10.1007/s10048-019-00572-7. Epub 2019 Mar 28. Neurogenetics. 2019. PMID: 30924036
-
Analysis of the Clinical Features and Imaging Findings of Pontocerebellar Hypoplasia Type 2D Caused by Mutations in SEPSECS Gene.Cerebellum. 2023 Oct;22(5):938-946. doi: 10.1007/s12311-022-01470-9. Epub 2022 Sep 9. Cerebellum. 2023. PMID: 36085396
-
Classification, diagnosis and potential mechanisms in pontocerebellar hypoplasia.Orphanet J Rare Dis. 2011 Jul 12;6:50. doi: 10.1186/1750-1172-6-50. Orphanet J Rare Dis. 2011. PMID: 21749694 Free PMC article. Review.
-
TSEN54 mutations cause pontocerebellar hypoplasia type 5.Eur J Hum Genet. 2011 Jun;19(6):724-6. doi: 10.1038/ejhg.2011.8. Epub 2011 Feb 2. Eur J Hum Genet. 2011. PMID: 21368912 Free PMC article.
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1007/BF01957367', 'is_inner': False, 'url': 'https://doi.org/10.1007/bf01957367'}, {'type': 'PubMed', 'value': '7607282', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7607282/'}]}
- Aalfs CM, van den Berg H, Barth PG, Hennekam RCM (1995) The Høyeraal-Hreidarsson syndrome: the fourth case of a separate entity with prenatal growth retardation, progressive pancytopenia and cerebellar hypoplasia. Eur J Pediatr 154:304–308 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1007/s00401-004-0975-3', 'is_inner': False, 'url': 'https://doi.org/10.1007/s00401-004-0975-3'}, {'type': 'PubMed', 'value': '15714316', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/15714316/'}]}
- Aronica E, van Kempen AA, van der Heide M, Poll-The BT, van Slooten HJ, Troost D, Rozemuller-Kwakkel JM (2005) Congenital disorder of glycosylation type Ia: a clinicopathological report of a newborn infant with cerebellar pathology. Acta Neuropathol (Berl) 109:433–442 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1007/BF00334450', 'is_inner': False, 'url': 'https://doi.org/10.1007/bf00334450'}, {'type': 'PubMed', 'value': '8480512', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/8480512/'}]}
- Albrecht S, Schneider MC, Belmont J, Armstrong DL (1993) Fatal infantile encephalopathy with olivopontocerebellar hypoplasia and micrencephaly. Acta Neuropathol 85:394–399 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '7791950', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7791950/'}]}
- Arts WFM, Hofstee Y, Drejer GF, Beverstock GC, Oosterwijk JC (1995) Cerebellar and brainstem hypoplasia in a child with a partial monosomy for the short arm of chromosome 5 and partial trisomy for the short arm of chromosome 10. Neuropediatrics 26:41–44 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1016/0022-510X(90)90096-6', 'is_inner': False, 'url': 'https://doi.org/10.1016/0022-510x(90)90096-6'}, {'type': 'PubMed', 'value': '2370559', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/2370559/'}]}
- Barth PG, Vrensen GFJM, Uylings HBM, Oorthuys JWE, Stam FC (1990) Inherited syndrome of microcephaly, dyskinesia and pontocerebellar hypoplasia: a systemic atrophy with early onset. J Neurol Sci 97:25–42 - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

