Thioredoxin-like domain of human kappa class glutathione transferase reveals sequence homology and structure similarity to the theta class enzyme
- PMID: 16081649
- PMCID: PMC2253485
- DOI: 10.1110/ps.051463905
Thioredoxin-like domain of human kappa class glutathione transferase reveals sequence homology and structure similarity to the theta class enzyme
Abstract
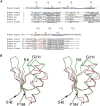
Glutathione transferases (GSTs) are a superfamily of enzymes that play a vital functional role in the cellular detoxification process. They catalyze the conjugation of the thiol group of glutathione (GSH) to the electrophilic groups of a wide range of hydrophobic substrates, leading to an easier removal of the latter from the cells. The kappa class is the least studied one among various classes within the superfamily. We report here the expression, purification, and crystal structure of human kappa class GST (hGSTK), which has been determined by the multiple-isomorphous replacement method and refined to 1.93 A resolution. The overall structure of hGSTK is similar to the recently reported structure of kappa class GST from rat mitochondrion. Each subunit of the dimeric hGSTK contains a thioredoxin (TRX)-like domain and a helical domain. A molecule of glutathione sulfinate, an oxidized product of GSH, is found to bind at the G site of each monomer. One oxygen atom of the sulfino group of GSF forms a hydrogen bond with the hydroxyl group of the catalytic residue Ser16. The TRX-like domain of hGSTK shares 19% sequence identity and structure similarity with human theta class GST, suggesting that the kappa class of GST is more closely related to the theta class enzyme within the GST superfamily. The structure of the TRX-like domain of hGSTK is also similar to that of glutathione peroxidase (GPx), implying an evolutionary relationship between GST and GPx.
Figures
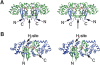
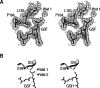
Similar articles
-
A mixed disulfide bond in bacterial glutathione transferase: functional and evolutionary implications.Structure. 1998 Jun 15;6(6):721-34. doi: 10.1016/s0969-2126(98)00074-4. Structure. 1998. PMID: 9655824
-
Crystal structures and kinetic studies of human Kappa class glutathione transferase provide insights into the catalytic mechanism.Biochem J. 2011 Oct 15;439(2):215-25. doi: 10.1042/BJ20110753. Biochem J. 2011. PMID: 21728995
-
Parallel evolutionary pathways for glutathione transferases: structure and mechanism of the mitochondrial class kappa enzyme rGSTK1-1.Biochemistry. 2004 Jan 20;43(2):352-61. doi: 10.1021/bi035832z. Biochemistry. 2004. PMID: 14717589
-
Structure, function and evolution of glutathione transferases: implications for classification of non-mammalian members of an ancient enzyme superfamily.Biochem J. 2001 Nov 15;360(Pt 1):1-16. doi: 10.1042/0264-6021:3600001. Biochem J. 2001. PMID: 11695986 Free PMC article. Review.
-
Mammalian class theta GST and differential susceptibility to carcinogens: a review.Mutat Res. 2000 Oct;463(3):247-83. doi: 10.1016/s1383-5742(00)00050-8. Mutat Res. 2000. PMID: 11018744 Review.
Cited by
-
Carvacrol, Thymol, and Garlic Essential Oil Promote Skin Innate Immunity in Gilthead Seabream (Sparus aurata) Through the Multifactorial Modulation of the Secretory Pathway and Enhancement of Mucus Protective Capacity.Front Immunol. 2021 Mar 12;12:633621. doi: 10.3389/fimmu.2021.633621. eCollection 2021. Front Immunol. 2021. PMID: 33777020 Free PMC article.
-
Functional Analysis of GSTK1 in Peroxisomal Redox Homeostasis in HEK-293 Cells.Antioxidants (Basel). 2023 Jun 7;12(6):1236. doi: 10.3390/antiox12061236. Antioxidants (Basel). 2023. PMID: 37371965 Free PMC article.
-
S-Glutathionyl-(chloro)hydroquinone reductases: a novel class of glutathione transferases.Biochem J. 2010 May 27;428(3):419-27. doi: 10.1042/BJ20091863. Biochem J. 2010. PMID: 20388120 Free PMC article.
-
Structures of a putative ζ-class glutathione S-transferase from the pathogenic fungus Coccidioides immitis.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011 Sep 1;67(Pt 9):1038-43. doi: 10.1107/S1744309111009493. Epub 2011 Aug 13. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2011. PMID: 21904047 Free PMC article.
-
ROS as Regulators of Cellular Processes in Melanoma.Oxid Med Cell Longev. 2021 Oct 23;2021:1208690. doi: 10.1155/2021/1208690. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 34725562 Free PMC article. Review.
References
-
- Armstrong, R.N. 1991. Glutathione S-transferases: Reaction mechanism, structure, and function. Chem. Res. Toxicol. 4 131–140. - PubMed
-
- Board, P.G., Coggan, M., Chelvanayagam, G., Easteal, S., Jermiin, L.S., Schulte, G.K., Danley, D.E., Hoth, L.R., Griffor, M.C., Kamath, A.V., et al. 2000. Identification, characterization, and crystal structure of the ω class glutathione transferases. J. Biol. Chem. 275 24798–24806. - PubMed
-
- Brünger, A.T., Adams, P.D., Clore, G.M., Delano, W.L., Gros, P., Grosse- Kunstleve, R.W., Jiang, J.-S., Kuszewski, J., Nilges, N., Pannu, N.S., et al. 1998. Crystallography and NMR systems: A new software system for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54 905–921. - PubMed
-
- Cameron, A.D., Sinning, I., L’Hermite, G., Olin, B., Board, P.G., Mannervik, B., and Jones, T.A. 1995. Structural analysis of human α-class glutathione transferase A1–1 in the apo-form and in complexes with ethacrynic acid and its glutathione conjugate. Structure 3 717–727. - PubMed
-
- Coles, B. and Ketterer, B. 1990. The role of glutathione and glutathione transferases in chemical carcinogenesis. Crit. Rev. Biochem.Mol. Biol. 25 47–70. - PubMed
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

