Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic
- PMID: 15647370
- PMCID: PMC545580
- DOI: 10.1073/pnas.0408864102
Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic
Abstract
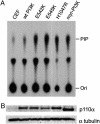
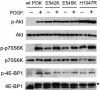
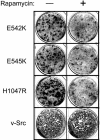
Mutations in genes that encode components of the phosphatidyl-inositol 3-kinase (PI3-kinase) signaling pathway are common in human cancer. The recent discovery of nonrandom somatic mutations in the PIK3CA gene of many human tumors suggests an oncogenic role for the mutated enzyme. We have determined the growth-regulatory and signaling properties of the three most frequently observed PI3-kinase mutations: E542K, E545K, and H1047R. Expressed in chicken embryo fibroblasts, all three mutants induce oncogenic transformation with high efficiency. This transforming ability is correlated with elevated catalytic activity in in vitro kinase assays. The mutant-transformed cells show constitutive phosphorylation of Akt, of p70 S6 kinase, and of the 4E-binding protein 1. Phosphorylation of S6 kinase and of 4E-binding protein 1 is regulated by the target of rapamycin (TOR) kinase and affects rates of protein synthesis. The inhibitor of TOR, rapamycin, strongly interferes with cellular transformation induced by the PI3-kinase mutants, suggesting that the TOR and its downstream targets are essential components of the transformation process. The oncogenic transforming activity makes the mutated PI3-kinase proteins promising targets for small molecule inhibitors that could be developed into effective and highly specific anticancer drugs.
Figures


Similar articles
-
A role of the kinase mTOR in cellular transformation induced by the oncoproteins P3k and Akt.Proc Natl Acad Sci U S A. 2001 Jan 2;98(1):136-41. doi: 10.1073/pnas.98.1.136. Proc Natl Acad Sci U S A. 2001. PMID: 11134523 Free PMC article.
-
Signal pathways involved in activation of p70S6K and phosphorylation of 4E-BP1 following exposure of multiple myeloma tumor cells to interleukin-6.J Biol Chem. 2002 May 3;277(18):15712-20. doi: 10.1074/jbc.M200043200. Epub 2002 Feb 28. J Biol Chem. 2002. PMID: 11872747
-
Constitutive activation of phosphatidyl-inositide 3 kinase contributes to the survival of Hodgkin's lymphoma cells through a mechanism involving Akt kinase and mTOR.J Pathol. 2005 Mar;205(4):498-506. doi: 10.1002/path.1725. J Pathol. 2005. PMID: 15714459
-
Retroviral oncogenes and TOR.Curr Top Microbiol Immunol. 2004;279:321-38. doi: 10.1007/978-3-642-18930-2_19. Curr Top Microbiol Immunol. 2004. PMID: 14560966 Review.
-
An essential role for protein synthesis in oncogenic cellular transformation.Oncogene. 2004 Apr 19;23(18):3145-50. doi: 10.1038/sj.onc.1207550. Oncogene. 2004. PMID: 15094764 Review.
Cited by
-
The PI3K/AKT Pathway and Renal Cell Carcinoma.J Genet Genomics. 2015 Jul 20;42(7):343-53. doi: 10.1016/j.jgg.2015.03.003. Epub 2015 Mar 19. J Genet Genomics. 2015. PMID: 26233890 Free PMC article. Review.
-
The pathogenesis of atypical proliferative Brenner tumor: an immunohistochemical and molecular genetic analysis.Mod Pathol. 2014 Feb;27(2):231-7. doi: 10.1038/modpathol.2013.142. Epub 2013 Jul 26. Mod Pathol. 2014. PMID: 23887305 Free PMC article.
-
Repression of p63 and induction of EMT by mutant Ras in mammary epithelial cells.Proc Natl Acad Sci U S A. 2016 Oct 11;113(41):E6107-E6116. doi: 10.1073/pnas.1613417113. Epub 2016 Sep 28. Proc Natl Acad Sci U S A. 2016. PMID: 27681615 Free PMC article.
-
Characterization of circulating breast cancer cells with tumorigenic and metastatic capacity.EMBO Mol Med. 2020 Sep 7;12(9):e11908. doi: 10.15252/emmm.201911908. Epub 2020 Jul 15. EMBO Mol Med. 2020. PMID: 32667137 Free PMC article.
-
Targeted agents in non-small cell lung cancer therapy: What is there on the horizon?J Carcinog. 2013 Mar 18;12:7. doi: 10.4103/1477-3163.109253. Print 2013. J Carcinog. 2013. PMID: 23599689 Free PMC article.
References
-
- Shayesteh, L., Lu, Y., Kuo, W. L., Baldocchi, R., Godfrey, T., Collins, C., Pinkel, D., Powell, B., Mills, G. B. & Gray, J. W. (1999) Nat. Genet. 21, 99-102. - PubMed
-
- Philp, A. J., Campbell, I. G., Leet, C., Vincan, E., Rockman, S. P., Whitehead, R. H., Thomas, R. J. & Phillips, W. A. (2001) Cancer Res. 61, 7426-7429. - PubMed
-
- Wang, S. I., Puc, J., Li, J., Bruce, J. N., Cairns, P., Sidransky, D. & Parsons, R. (1997) Cancer Res. 57, 4183-4186. - PubMed
-
- Yokoyama, Y., Wan, X., Shinohara, A., Takahashi, S., Takahashi, Y., Niwa, K. & Tamaya, T. (2000) Int. J. Mol. Med. 6, 47-50. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

