TGF-beta-dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome
- PMID: 15546004
- PMCID: PMC529498
- DOI: 10.1172/JCI22715
TGF-beta-dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome
Abstract
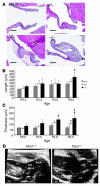
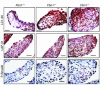
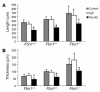
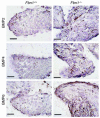
Mitral valve prolapse (MVP) is a common human phenotype, yet little is known about the pathogenesis of this condition. MVP can occur in the context of genetic syndromes, including Marfan syndrome (MFS), an autosomal-dominant connective tissue disorder caused by mutations in fibrillin-1. Fibrillin-1 contributes to the regulated activation of the cytokine TGF-beta, and enhanced signaling is a consequence of fibrillin-1 deficiency. We thus hypothesized that increased TGF-beta signaling may contribute to the multisystem pathogenesis of MFS, including the development of myxomatous changes of the atrioventricular valves. Mitral valves from fibrillin-1-deficient mice exhibited postnatally acquired alterations in architecture that correlated both temporally and spatially with increased cell proliferation, decreased apoptosis, and excess TGF-beta activation and signaling. In addition, TGF-beta antagonism in vivo rescued the valve phenotype, suggesting a cause and effect relationship. Expression analyses identified increased expression of numerous TGF-beta-related genes that regulate cell proliferation and survival and plausibly contribute to myxomatous valve disease. These studies validate a novel, genetically engineered murine model of myxomatous changes of the mitral valve and provide critical insight into the pathogenetic mechanism of such changes in MFS and perhaps more common nonsyndromic variants of mitral valve disease.
Figures

Comment in
-
Marfan syndrome and mitral valve prolapse.J Clin Invest. 2004 Dec;114(11):1543-6. doi: 10.1172/JCI23701. J Clin Invest. 2004. PMID: 15578086 Free PMC article.
Similar articles
-
Marfan syndrome and mitral valve prolapse.J Clin Invest. 2004 Dec;114(11):1543-6. doi: 10.1172/JCI23701. J Clin Invest. 2004. PMID: 15578086 Free PMC article.
-
Mitral valve disease in Marfan syndrome and related disorders.J Cardiovasc Transl Res. 2011 Dec;4(6):741-7. doi: 10.1007/s12265-011-9314-y. Epub 2011 Aug 25. J Cardiovasc Transl Res. 2011. PMID: 21866385 Review.
-
Dysregulation of TGF-beta activation contributes to pathogenesis in Marfan syndrome.Nat Genet. 2003 Mar;33(3):407-11. doi: 10.1038/ng1116. Epub 2003 Feb 24. Nat Genet. 2003. PMID: 12598898
-
miR-29b participates in early aneurysm development in Marfan syndrome.Circ Res. 2012 Jan 20;110(2):312-24. doi: 10.1161/CIRCRESAHA.111.253740. Epub 2011 Nov 23. Circ Res. 2012. PMID: 22116819
-
Fibrillin in Marfan syndrome and tight skin mice provides new insights into transforming growth factor-beta regulation and systemic sclerosis.Curr Opin Rheumatol. 2006 Nov;18(6):582-7. doi: 10.1097/01.bor.0000245719.64393.57. Curr Opin Rheumatol. 2006. PMID: 17053502 Review.
Cited by
-
Myxomatous mitral valve disease in dogs: does size matter?J Vet Cardiol. 2012 Mar;14(1):19-29. doi: 10.1016/j.jvc.2012.01.006. Epub 2012 Feb 20. J Vet Cardiol. 2012. PMID: 22356836 Free PMC article. Review.
-
Cardiac Embryology and Molecular Mechanisms of Congenital Heart Disease: A Primer for Anesthesiologists.Anesth Analg. 2016 Sep;123(3):551-69. doi: 10.1213/ANE.0000000000001451. Anesth Analg. 2016. PMID: 27541719 Free PMC article. Review.
-
TGF-β signalling and reactive oxygen species drive fibrosis and matrix remodelling in myxomatous mitral valves.Cardiovasc Res. 2013 Jul 1;99(1):175-84. doi: 10.1093/cvr/cvt083. Epub 2013 Apr 3. Cardiovasc Res. 2013. PMID: 23554457 Free PMC article.
-
Educational paper. Connective tissue disorders with vascular involvement: from gene to therapy.Eur J Pediatr. 2013 Aug;172(8):997-1005. doi: 10.1007/s00431-012-1773-x. Epub 2012 Jul 17. Eur J Pediatr. 2013. PMID: 22801769 Free PMC article. Review.
-
Biglycan and fibromodulin have essential roles in regulating chondrogenesis and extracellular matrix turnover in temporomandibular joint osteoarthritis.Am J Pathol. 2010 Feb;176(2):812-26. doi: 10.2353/ajpath.2010.090450. Epub 2009 Dec 24. Am J Pathol. 2010. PMID: 20035055 Free PMC article.
References
-
- Pyeritz RE. The Marfan syndrome. Annu. Rev. Med. 2000;51:481–510. - PubMed
-
- Dietz HC, et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature. 1991;352:337–339. - PubMed
-
- Tamura K, et al. Abnormalities in elastic fibers and other connective-tissue components of floppy mitral valve. Am. Heart J. 1995;129:1149–1158. - PubMed
-
- Durbin AD, Gotlieb AI. Advances towards understanding heart valve response to injury. Cardiovasc. Pathol. 2002;11:69–77. - PubMed
-
- Weber H, et al. Endothelin-1 and angiotensin-II stimulate delayed mitogenesis in cultured rat aortic smooth muscle cells: evidence for common signaling mechanisms. Mol. Endocrinol. 1994;8:148–158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

