TodK, a putative histidine protein kinase, regulates timing of fruiting body morphogenesis in Myxococcus xanthus
- PMID: 12949097
- PMCID: PMC193762
- DOI: 10.1128/JB.185.18.5452-5464.2003
TodK, a putative histidine protein kinase, regulates timing of fruiting body morphogenesis in Myxococcus xanthus
Abstract
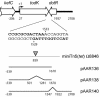
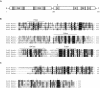
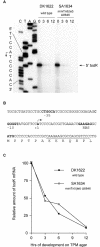
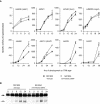
In response to starvation, Myxococcus xanthus initiates a developmental program that results in the formation of spore-filled multicellular fruiting bodies. Fruiting body formation depends on the temporal and spatial coordination of aggregation and sporulation. These two processes are induced by the cell surface-associated C signal, with aggregation being induced after 6 h and sporulation being induced once cells have completed the aggregation process. We report the identification of TodK, a putative histidine protein kinase of two-component regulatory systems that is important for the correct timing of aggregation and sporulation. Loss of TodK function results in early aggregation and early, as well as increased levels of, sporulation. Transcription of todK decreases 10-fold in response to starvation independently of the stringent response. Loss of TodK function specifically results in increased expression of a subset of C-signal-dependent genes. Accelerated development in a todK mutant depends on the known components in the C-signal transduction pathway. TodK is not important for synthesis of the C signal. From these results we suggest that TodK is part of a signal transduction system which converges on the C-signal transduction pathway to negatively regulate aggregation, sporulation, and the expression of a subset of C-signal-dependent genes. TodK and the SdeK histidine protein kinase, which is part of a signal transduction system that converges on the C-signal transduction pathway to stimulate aggregation, sporulation, and C-signal-dependent gene expression, act in independent genetic pathways. We suggest that the signal transduction pathways defined by TodK and SdeK act in concert with the C-signal transduction pathway to control the timing of aggregation and sporulation.
Figures


Similar articles
-
Genetic studies of mrp, a locus essential for cellular aggregation and sporulation of Myxococcus xanthus.J Bacteriol. 2001 Aug;183(16):4786-95. doi: 10.1128/JB.183.16.4786-4795.2001. J Bacteriol. 2001. PMID: 11466282 Free PMC article.
-
HthA, a putative DNA-binding protein, and HthB are important for fruiting body morphogenesis in Myxococcus xanthus.Microbiology (Reading). 2004 Jul;150(Pt 7):2171-2183. doi: 10.1099/mic.0.27151-0. Microbiology (Reading). 2004. PMID: 15256560
-
Myxococcus xanthus sasS encodes a sensor histidine kinase required for early developmental gene expression.J Bacteriol. 1997 Dec;179(24):7759-67. doi: 10.1128/jb.179.24.7759-7767.1997. J Bacteriol. 1997. PMID: 9401035 Free PMC article.
-
Dual regulation with Ser/Thr kinase cascade and a His/Asp TCS in Myxococcus xanthus.Adv Exp Med Biol. 2008;631:111-21. doi: 10.1007/978-0-387-78885-2_7. Adv Exp Med Biol. 2008. PMID: 18792684 Review.
-
Two-Component Signal Transduction Systems That Regulate the Temporal and Spatial Expression of Myxococcus xanthus Sporulation Genes.J Bacteriol. 2015 Sep 14;198(3):377-85. doi: 10.1128/JB.00474-15. Print 2016 Feb 1. J Bacteriol. 2015. PMID: 26369581 Free PMC article. Review.
Cited by
-
Myxococcus xanthus fruiting body morphology is important for spore recovery after exposure to environmental stress.Appl Environ Microbiol. 2024 Oct 23;90(10):e0166024. doi: 10.1128/aem.01660-24. Epub 2024 Oct 4. Appl Environ Microbiol. 2024. PMID: 39365039
-
Global transcriptome analysis of spore formation in Myxococcus xanthus reveals a locus necessary for cell differentiation.BMC Genomics. 2010 Apr 26;11:264. doi: 10.1186/1471-2164-11-264. BMC Genomics. 2010. PMID: 20420673 Free PMC article.
-
Intra- and interprotein phosphorylation between two-hybrid histidine kinases controls Myxococcus xanthus developmental progression.J Biol Chem. 2012 Jul 20;287(30):25060-72. doi: 10.1074/jbc.M112.387241. Epub 2012 Jun 1. J Biol Chem. 2012. PMID: 22661709 Free PMC article.
-
EspA, an orphan hybrid histidine protein kinase, regulates the timing of expression of key developmental proteins of Myxococcus xanthus.J Bacteriol. 2008 Jul;190(13):4416-26. doi: 10.1128/JB.00265-08. Epub 2008 Apr 4. J Bacteriol. 2008. PMID: 18390653 Free PMC article.
-
A novel "four-component" two-component signal transduction mechanism regulates developmental progression in Myxococcus xanthus.J Biol Chem. 2009 Aug 7;284(32):21435-45. doi: 10.1074/jbc.M109.033415. Epub 2009 Jun 17. J Biol Chem. 2009. PMID: 19535336 Free PMC article.
References
-
- Altschul, S. F., W. Gish, W. Miller, E. W. Myers, and D. J. Lipman. 1990. Basic local alignment search tool. J. Mol. Biol. 215:403-410. - PubMed
-
- Bibb, M. J., P. R. Findlay, and M. W. Johnson. 1984. The relationship between base composition and codon usage in bacterial genes and its use for the simple and reliable identification of protein-coding sequences. Gene 30:157-166. - PubMed
-
- Cho, K., and D. R. Zusman. 1999. AsgD, a new two-component regulator required for A-signalling and nutrient sensing during early development of Myxococcus xanthus. Mol. Microbiol. 34:268-281. - PubMed
-
- Cho, K., and D. R. Zusman. 1999. Sporulation timing in Myxococcus xanthus is controlled by the espAB locus. Mol. Microbiol. 34:714-725. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources

