Microtubule-associated protein 1B: a neuronal binding partner for gigaxonin
- PMID: 12147674
- PMCID: PMC2173828
- DOI: 10.1083/jcb.200202055
Microtubule-associated protein 1B: a neuronal binding partner for gigaxonin
Erratum in
- J Cell Biol. 2003 Oct 13;163(1):189
Abstract
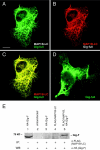
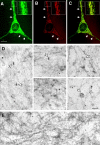
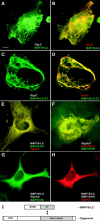
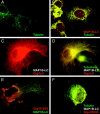
Giant axonal neuropathy (GAN), an autosomal recessive disorder caused by mutations in GAN, is characterized cytopathologically by cytoskeletal abnormality. Based on its sequence, gigaxonin contains an NH2-terminal BTB domain followed by six kelch repeats, which are believed to be important for protein-protein interactions (Adams, J., R. Kelso, and L. Cooley. 2000. Trends Cell Biol. 10:17-24.). Here, we report the identification of a neuronal binding partner of gigaxonin. Results obtained from yeast two-hybrid screening, cotransfections, and coimmunoprecipitations demonstrate that gigaxonin binds directly to microtubule-associated protein (MAP)1B light chain (LC; MAP1B-LC), a protein involved in maintaining the integrity of cytoskeletal structures and promoting neuronal stability. Studies using double immunofluorescent microscopy and ultrastructural analysis revealed physiological colocalization of gigaxonin with MAP1B in neurons. Furthermore, in transfected cells the specific interaction of gigaxonin with MAP1B is shown to enhance the microtubule stability required for axonal transport over long distance. At least two different mutations identified in GAN patients (Bomont, P., L. Cavalier, F. Blondeau, C. Ben Hamida, S. Belal, M. Tazir, E. Demir, H. Topaloglu, R. Korinthenberg, B. Tuysuz, et al. 2000. Nat. Genet. 26:370-374.) lead to loss of gigaxonin-MAP1B-LC interaction. The devastating axonal degeneration and neuronal death found in GAN patients point to the importance of gigaxonin for neuronal survival. Our findings may provide important insights into the pathogenesis of neurodegenerative disorders related to cytoskeletal abnormalities.
Figures

Similar articles
-
Gigaxonin-controlled degradation of MAP1B light chain is critical to neuronal survival.Nature. 2005 Nov 10;438(7065):224-8. doi: 10.1038/nature04256. Epub 2005 Oct 16. Nature. 2005. PMID: 16227972
-
The gene encoding gigaxonin, a new member of the cytoskeletal BTB/kelch repeat family, is mutated in giant axonal neuropathy.Nat Genet. 2000 Nov;26(3):370-4. doi: 10.1038/81701. Nat Genet. 2000. PMID: 11062483
-
Gene targeting of GAN in mouse causes a toxic accumulation of microtubule-associated protein 8 and impaired retrograde axonal transport.Hum Mol Genet. 2006 May 1;15(9):1451-63. doi: 10.1093/hmg/ddl069. Epub 2006 Mar 24. Hum Mol Genet. 2006. PMID: 16565160
-
Microtubule-associated protein 1B, a growth-associated and phosphorylated scaffold protein.Brain Res Bull. 2007 Mar 30;71(6):541-58. doi: 10.1016/j.brainresbull.2006.11.012. Epub 2006 Dec 27. Brain Res Bull. 2007. PMID: 17292797 Review.
-
Giant axonal neuropathy.Handb Clin Neurol. 2013;115:933-8. doi: 10.1016/B978-0-444-52902-2.00052-7. Handb Clin Neurol. 2013. PMID: 23931822 Review.
Cited by
-
Nemitin, a novel Map8/Map1s interacting protein with Wd40 repeats.PLoS One. 2012;7(4):e33094. doi: 10.1371/journal.pone.0033094. Epub 2012 Apr 16. PLoS One. 2012. PMID: 22523538 Free PMC article.
-
Tubulin Folding Cofactor TBCB is a Target of the Salmonella Effector Protein SseK1.Int J Mol Sci. 2020 Apr 30;21(9):3193. doi: 10.3390/ijms21093193. Int J Mol Sci. 2020. PMID: 32366039 Free PMC article.
-
Review of the multiple aspects of neurofilament functions, and their possible contribution to neurodegeneration.Mol Neurobiol. 2008 Aug;38(1):27-65. doi: 10.1007/s12035-008-8033-0. Epub 2008 Jul 23. Mol Neurobiol. 2008. PMID: 18649148 Review.
-
Gigaxonin controls vimentin organization through a tubulin chaperone-independent pathway.Hum Mol Genet. 2009 Apr 15;18(8):1384-94. doi: 10.1093/hmg/ddp044. Epub 2009 Jan 24. Hum Mol Genet. 2009. PMID: 19168853 Free PMC article.
-
A review of gigaxonin mutations in giant axonal neuropathy (GAN) and cancer.Hum Genet. 2016 Jul;135(7):675-84. doi: 10.1007/s00439-016-1659-5. Epub 2016 Mar 29. Hum Genet. 2016. PMID: 27023907 Review.
References
-
- Adams, J., R. Kelso, and L. Cooley. 2000. The kelch repeat superfamily of proteins: propellers of cell function. Trends Cell Biol. 10:17–24. - PubMed
-
- Ben Hamída, C., L. Cavalier, S. Belal, H. Sanhaji, N. Nadal, C. Barhoumi, N. M'Rissa, N. Marzouki, J.L. Mandel, M. Ben Hamída, et al. 1997. Homozygosity mapping of giant axonal neuropathy gene to chromosome 16q24.1. Neurogenetics. 1:129–133. - PubMed
-
- Berg, B.O., S.H. Rosenberg, and A.K. Asbury. 1972. Giant axonal neuropathy. Pediatrics. 49:894–899. - PubMed
-
- Bomont, P., L. Cavalier, F. Blondeau, C. Ben Hamida, S. Belal, M. Tazir, E. Demir, H. Topaloglu, R. Korinthenberg, B. Tuysuz, et al. 2000. The gene encoding gigaxonin, a new member of the cytoskeletal BTB/kelch repeat family, is mutated in giant axonal neuropathy. Nat. Genet. 26:370–374. - PubMed
-
- Bousquet, O., M. Basseville, E. Vila-Porcile, T. Billette de Villemeur, J.J. Hauw, P. Landrieu, and M.M. Portier. 1996. Aggregation of a subpopulation of vimentin filaments in cultured human skin fibroblasts derived from patients with giant axonal neuropathy. Cell Motil. Cytoskeleton. 33:115–129. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

