Lysine methylation within the globular domain of histone H3 by Dot1 is important for telomeric silencing and Sir protein association
- PMID: 12080090
- PMCID: PMC186335
- DOI: 10.1101/gad.1001502
Lysine methylation within the globular domain of histone H3 by Dot1 is important for telomeric silencing and Sir protein association
Abstract
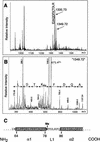
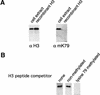
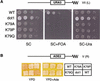
The amino-terminal histone tails are subject to covalent post-translational modifications such as acetylation, methylation, and phosphorylation. In the histone code hypothesis, these exposed and unstructured histone tails are accessible to a repertoire of regulatory factors that specifically recognize the various modified histones, thereby generating altered chromatin structures that mediate specific biological responses. Here, we report that lysine (Lys) 79 of histone H3, which resides in the globular domain, is methylated in eukaryotic organisms. In the yeast Saccharomyces cerevisiae, Lys 79 of histone H3 is methylated by Dot1, a protein shown previously to play a role in telomeric silencing. Mutations of Lys 79 of histone H3 and mutations that abolish the catalytic activity of Dot1 impair telomeric silencing, suggesting that Dot1 mediates telomeric silencing largely through methylation of Lys 79. This defect in telomeric silencing might reflect an interaction between Sir proteins and Lys 79, because dot1 and Lys 79 mutations weaken the interaction of Sir2 and Sir3 with the telomeric region in vivo. Our results indicate that histone modifications in the core globular domain have important biological functions.
Figures



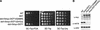


Similar articles
-
Rap1-Sir4 binding independent of other Sir, yKu, or histone interactions initiates the assembly of telomeric heterochromatin in yeast.Genes Dev. 2002 Jun 15;16(12):1528-39. doi: 10.1101/gad.988802. Genes Dev. 2002. PMID: 12080091 Free PMC article.
-
Evidence that Set1, a factor required for methylation of histone H3, regulates rDNA silencing in S. cerevisiae by a Sir2-independent mechanism.Curr Biol. 2002 Jan 22;12(2):165-70. doi: 10.1016/s0960-9822(01)00652-2. Curr Biol. 2002. PMID: 11818070
-
Histone H3 lysine 36 methylation antagonizes silencing in Saccharomyces cerevisiae independently of the Rpd3S histone deacetylase complex.Genetics. 2007 Feb;175(2):585-93. doi: 10.1534/genetics.106.067751. Epub 2006 Dec 18. Genetics. 2007. PMID: 17179083 Free PMC article.
-
The many faces of histone H3K79 methylation.Mutat Res Rev Mutat Res. 2016 Apr-Jun;768:46-52. doi: 10.1016/j.mrrev.2016.03.005. Epub 2016 Mar 31. Mutat Res Rev Mutat Res. 2016. PMID: 27234562 Free PMC article. Review.
-
The Sir proteins of Saccharomyces cerevisiae: mediators of transcriptional silencing and much more.Curr Opin Microbiol. 2000 Apr;3(2):132-7. doi: 10.1016/s1369-5274(00)00064-3. Curr Opin Microbiol. 2000. PMID: 10744999 Review.
Cited by
-
Epigenetics of the failing heart.Heart Fail Rev. 2015 Jul;20(4):435-59. doi: 10.1007/s10741-015-9483-x. Heart Fail Rev. 2015. PMID: 25847519 Review.
-
Characterization of the grappa gene, the Drosophila histone H3 lysine 79 methyltransferase.Genetics. 2005 Jan;169(1):173-84. doi: 10.1534/genetics.104.033191. Epub 2004 Sep 15. Genetics. 2005. PMID: 15371351 Free PMC article.
-
Histone Methyltransferases as Therapeutic Targets for Kidney Diseases.Front Pharmacol. 2019 Dec 6;10:1393. doi: 10.3389/fphar.2019.01393. eCollection 2019. Front Pharmacol. 2019. PMID: 31866860 Free PMC article. Review.
-
Sensitive ChIP-DSL technology reveals an extensive estrogen receptor alpha-binding program on human gene promoters.Proc Natl Acad Sci U S A. 2007 Mar 20;104(12):4852-7. doi: 10.1073/pnas.0700715104. Epub 2007 Mar 14. Proc Natl Acad Sci U S A. 2007. PMID: 17360330 Free PMC article.
-
Using Yeast to Define the Regulatory Role of Protein Lysine Methylation.Curr Protein Pept Sci. 2020;21(7):690-698. doi: 10.2174/1389203720666191023150727. Curr Protein Pept Sci. 2020. PMID: 31642774 Free PMC article. Review.
References
-
- Aparicio OM, Billington BL, Gottschling DE. Modifiers of position effect are shared between telomeric and silent mating-type loci in S. cerevisiae. Cell. 1991;66:1279–1287. - PubMed
-
- Bannister AJ, Zegerman P, Partridge JF, Miska EA, Thomas JO, Allshire RC, Kouzarides T. Selective recognition of methylated lysine 9 on histone H3 by the HP1 chromo domain. Nature. 2001;410:120–124. - PubMed
-
- Boggs BA, Cheung P, Heard E, Spector DL, Chinault AC, Allis CD. Differentially methylated forms of histone H3 show unique association patterns with inactive human X chromosomes. Nat Genet. 2002;30:73–76. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
