Targeted deletion of both thymidine phosphorylase and uridine phosphorylase and consequent disorders in mice
- PMID: 12077348
- PMCID: PMC139774
- DOI: 10.1128/MCB.22.14.5212-5221.2002
Targeted deletion of both thymidine phosphorylase and uridine phosphorylase and consequent disorders in mice
Abstract
Thymidine phosphorylase (TP) regulates intracellular and plasma thymidine levels. TP deficiency is hypothesized to (i) increase levels of thymidine in plasma, (ii) lead to mitochondrial DNA alterations, and (iii) cause mitochondrial neurogastrointestinal encephalomyopathy (MNGIE). In order to elucidate the physiological roles of TP, we generated mice deficient in the TP gene. Although TP activity in the liver was inhibited in these mice, it was fully maintained in the small intestine. Murine uridine phosphorylase (UP), unlike human UP, cleaves thymidine, as well as uridine. We therefore generated TP-UP double-knockout (TP(-/-) UP(-/-)) mice. TP activities were inhibited in TP(-/-) UP(-/-) mice, and the level of thymidine in the plasma of TP(-/-) UP(-/-) mice was higher than for TP(-/-) mice. Unexpectedly, we could not observe alterations of mitochondrial DNA or pathological changes in the muscles of the TP(-/-) UP(-/-) mice, even when these mice were fed thymidine for 7 months. However, we did find hyperintense lesions on magnetic resonance T(2) maps in the brain and axonal edema by electron microscopic study of the brain in TP(-/-) UP(-/-) mice. These findings suggested that the inhibition of TP activity caused the elevation of pyrimidine levels in plasma and consequent axonal swelling in the brains of mice. Since lesions in the brain do not appear to be due to mitochondrial alterations and pathological changes in the muscle were not found, this model will provide further insights into the causes of MNGIE.
Figures


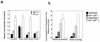


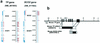
Similar articles
-
Unbalanced deoxynucleotide pools cause mitochondrial DNA instability in thymidine phosphorylase-deficient mice.Hum Mol Genet. 2009 Feb 15;18(4):714-22. doi: 10.1093/hmg/ddn401. Epub 2008 Nov 21. Hum Mol Genet. 2009. PMID: 19028666 Free PMC article.
-
Deoxynucleoside stress exacerbates the phenotype of a mouse model of mitochondrial neurogastrointestinal encephalopathy.Brain. 2014 May;137(Pt 5):1337-49. doi: 10.1093/brain/awu068. Epub 2014 Apr 10. Brain. 2014. PMID: 24727567 Free PMC article.
-
Long-Term Restoration of Thymidine Phosphorylase Function and Nucleoside Homeostasis Using Hematopoietic Gene Therapy in a Murine Model of Mitochondrial Neurogastrointestinal Encephalomyopathy.Hum Gene Ther. 2016 Sep;27(9):656-67. doi: 10.1089/hum.2015.160. Epub 2016 May 4. Hum Gene Ther. 2016. PMID: 27004974 Free PMC article.
-
Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): a disease of two genomes.Neurologist. 2004 Jan;10(1):8-17. doi: 10.1097/01.nrl.0000106919.06469.04. Neurologist. 2004. PMID: 14720311 Review.
-
MNGIE: from nuclear DNA to mitochondrial DNA.Neuromuscul Disord. 2001 Jan;11(1):7-10. doi: 10.1016/s0960-8966(00)00159-0. Neuromuscul Disord. 2001. PMID: 11166160 Review.
Cited by
-
Animal Models of Mitochondrial Diseases Associated with Nuclear Gene Mutations.Acta Naturae. 2023 Oct-Dec;15(4):4-22. doi: 10.32607/actanaturae.25442. Acta Naturae. 2023. PMID: 38234606 Free PMC article.
-
A discovery resource of rare copy number variations in individuals with autism spectrum disorder.G3 (Bethesda). 2012 Dec;2(12):1665-85. doi: 10.1534/g3.112.004689. Epub 2012 Dec 1. G3 (Bethesda). 2012. PMID: 23275889 Free PMC article.
-
Mitochondria.J Neurol Neurosurg Psychiatry. 2003 Sep;74(9):1188-99. doi: 10.1136/jnnp.74.9.1188. J Neurol Neurosurg Psychiatry. 2003. PMID: 12933917 Free PMC article. Review.
-
Unbalanced deoxynucleotide pools cause mitochondrial DNA instability in thymidine phosphorylase-deficient mice.Hum Mol Genet. 2009 Feb 15;18(4):714-22. doi: 10.1093/hmg/ddn401. Epub 2008 Nov 21. Hum Mol Genet. 2009. PMID: 19028666 Free PMC article.
-
Modeling pathogenic mutations of human twinkle in Drosophila suggests an apoptosis role in response to mitochondrial defects.PLoS One. 2012;7(8):e43954. doi: 10.1371/journal.pone.0043954. Epub 2012 Aug 28. PLoS One. 2012. PMID: 22952820 Free PMC article.
References
-
- Carrozzo, R., M. Hirano, B. Fromenty, C. Casali, F. M. Santorelli, E. Bonilla, S. DiMauro, E. A. Schon, and A. F. Miranda. 1998. Multiple mtDNA deletions features in autosomal dominant and recessive diseases suggest distinct pathogeneses. Neurology 50:99-106. - PubMed
-
- Debouverie, M., M. Wagner, X. Ducrocq, Y. Grignon, B. Mousson, and M. Weber. 1997. MNGIE syndrome in two siblings. Rev. Neurol. 153:547-553. - PubMed
-
- Dickinson, E. K., D. L. Adams, E. A. Schon, and D. M. Glerum. 2000. A human SCO2 mutation helps define the role of Sco1p in the cytochrome oxidase assembly pathway. J. Biol. Chem. 275:26780-26785. - PubMed
-
- elKouni, M. H., M. M. elKouni, and F. N. Naguib. 1993. Differences in activities and substrate specificity of human and murine pyrimidine nucleoside phosphorylases: implications for chemotherapy with 5-fluoropyrimidines. Cancer Res. 53:3687-3693. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
