Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein
- PMID: 11179006
- PMCID: PMC1274471
- DOI: 10.1086/318811
Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein
Abstract
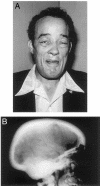
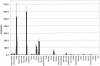
Sclerosteosis is an autosomal recessive sclerosing bone dysplasia characterized by progressive skeletal overgrowth. The majority of affected individuals have been reported in the Afrikaner population of South Africa, where a high incidence of the disorder occurs as a result of a founder effect. Homozygosity mapping in Afrikaner families along with analysis of historical recombinants localized sclerosteosis to an interval of approximately 2 cM between the loci D17S1787 and D17S930 on chromosome 17q12-q21. Here we report two independent mutations in a novel gene, termed "SOST." Affected Afrikaners carry a nonsense mutation near the amino terminus of the encoded protein, whereas an unrelated affected person of Senegalese origin carries a splicing mutation within the single intron of the gene. The SOST gene encodes a protein that shares similarity with a class of cystine knot-containing factors including dan, cerberus, gremlin, prdc, and caronte. The specific and progressive effect on bone formation observed in individuals affected with sclerosteosis, along with the data presented in this study, together suggest that the SOST gene encodes an important new regulator of bone homeostasis.
Figures
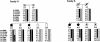


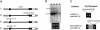


Similar articles
-
Increased bone density in sclerosteosis is due to the deficiency of a novel secreted protein (SOST).Hum Mol Genet. 2001 Mar 1;10(5):537-43. doi: 10.1093/hmg/10.5.537. Hum Mol Genet. 2001. PMID: 11181578
-
Sclerosteosis caused by a novel nonsense mutation of SOST in a consanguineous family.Clin Genet. 2016 Feb;89(2):205-9. doi: 10.1111/cge.12655. Epub 2015 Sep 22. Clin Genet. 2016. PMID: 26283468
-
Identification of a 52 kb deletion downstream of the SOST gene in patients with van Buchem disease.J Med Genet. 2002 Feb;39(2):91-7. doi: 10.1136/jmg.39.2.91. J Med Genet. 2002. PMID: 11836356 Free PMC article.
-
Genetics of Sost/SOST in sclerosteosis and van Buchem disease animal models.Metabolism. 2018 Mar;80:38-47. doi: 10.1016/j.metabol.2017.10.005. Epub 2017 Oct 25. Metabolism. 2018. PMID: 29080811 Review.
-
Identification of the disease-causing gene in sclerosteosis--discovery of a novel bone anabolic target?J Musculoskelet Neuronal Interact. 2004 Jun;4(2):139-42. J Musculoskelet Neuronal Interact. 2004. PMID: 15615113 Review.
Cited by
-
Only minor differences in renal osteodystrophy features between wild-type and sclerostin knockout mice with chronic kidney disease.Kidney Int. 2016 Oct;90(4):828-34. doi: 10.1016/j.kint.2016.06.019. Epub 2016 Aug 12. Kidney Int. 2016. PMID: 27528549 Free PMC article.
-
Sclerostin antibody treatment improves fracture outcomes in a Type I diabetic mouse model.Bone. 2016 Jan;82:122-34. doi: 10.1016/j.bone.2015.04.048. Epub 2015 May 5. Bone. 2016. PMID: 25952969 Free PMC article.
-
Targeting Wnt pathways in disease.Cold Spring Harb Perspect Biol. 2012 Nov 1;4(11):a008086. doi: 10.1101/cshperspect.a008086. Cold Spring Harb Perspect Biol. 2012. PMID: 23001988 Free PMC article. Review.
-
Lactating Ctcgrp nulls lose twice the normal bone mineral content due to fewer osteoblasts and more osteoclasts, whereas bone mass is fully restored after weaning in association with up-regulation of Wnt signaling and other novel genes.Endocrinology. 2013 Apr;154(4):1400-13. doi: 10.1210/en.2012-1931. Epub 2013 Mar 5. Endocrinology. 2013. PMID: 23462960 Free PMC article.
-
Bone metabolism in patients with anorexia nervosa and amenorrhoea.Eat Weight Disord. 2018 Apr;23(2):255-261. doi: 10.1007/s40519-016-0337-x. Epub 2016 Oct 27. Eat Weight Disord. 2018. PMID: 27787773
References
Electronic-Database Information
-
- CEPH, http://www.cephb.fr/
-
- Cooperative Human Linkage Center, http://lpg.nci.nih.gov/CHLC/
-
- Coriell Cell Repositories, http://locus.umdnj.edu/
-
- Expressed Sequence Tags database, http://www.ncbi.nlm.nih.gov/dbEST/index.html
References
-
- Balemans W, van den Ende J, Freire Paes-Alves A, Dikkers F, Willems P, Vanhoenacker F, de Almeida-Melo N, Alves CF, Stratakis CA, Hill SC, van Hul W (1999) Localization of the gene for sclerosteosis to the van Buchem disease-gene region on chromosome 17q12-q21. Am J Hum Genet 64:1661–1669 - PMC - PubMed
-
- Beighton P, Barnard A, Hamersma H, van der Wouden A (1984) The syndromic status of sclerosteosis and van Buchem disease. Clin Genet 25:175–181 - PubMed
-
- Beighton P, Davidson J, Durr L, Hamersma H (1977) Sclerosteosis—an autosomal recessive disorder. Clin Genet 11:1–7 - PubMed
-
- Beighton P, Durr L, Hamersma H (1976) The clinical features of sclerosteosis: a review of the manifestations in twenty-five affected individuals. Ann Intern Med 84:393–397 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

