Multiplex Western blotting system for the analysis of muscular dystrophy proteins
- PMID: 10233840
- PMCID: PMC1866550
- DOI: 10.1016/S0002-9440(10)65354-0
Multiplex Western blotting system for the analysis of muscular dystrophy proteins
Abstract
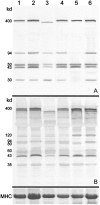
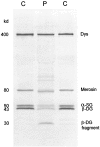
A multiplex system of Western blotting is presented in which most of the current muscular dystrophy proteins can be analyzed simultaneously on one pair of blots. This represents a significant improvement in efficiency and cost for this type of analysis. The final diagnosis is more quickly achieved in patients where several possible diagnoses are indicated after clinical appraisal, and those with unusual presentations may be quickly resolved. The method uses a biphasic polyacrylamide gel system, which enables the corresponding blot to be probed simultaneously with a cocktail of monoclonal antibodies. The gel is optimized so that large proteins of more than 200 kd (eg, dystrophin, dysferlin, and myosin heavy chain) can be analyzed in the top part, while smaller proteins under 150 kd (eg, calpain 3, the 80-kd fragment of laminin alpha2 chain, all of the sarcoglycans, and caveolin 3) are separated in the lower phase. This basic system could be used for different combinations of antibodies as new muscular dystrophy proteins are identified and require examination. In addition, analysis of the laminin alpha2 chain of merosin showed that this protein was expressed as a doublet or triplet set of bands in many patients with active muscle pathology. This may indicate the existence of an embryonic isoform, which is re-expressed in regenerating fibers.
Figures



Similar articles
-
Altered distribution of beta-dystroglycan in sarcolemma of human dystrophic muscles: an immunohistochemical study.Muscle Nerve. 1995 Aug;18(8):910-3. doi: 10.1002/mus.880180818. Muscle Nerve. 1995. PMID: 7630355 No abstract available.
-
Abnormalities of dystrophin, the sarcoglycans, and laminin alpha2 in the muscular dystrophies.J Med Genet. 1998 May;35(5):379-86. doi: 10.1136/jmg.35.5.379. J Med Genet. 1998. PMID: 9610800 Free PMC article.
-
[Dystrophinopathies, congenital muscular dystrophy, limb-girdle dystrophies: updated classification].Rev Neurol. 1998 Jun;26(154):1021-6. Rev Neurol. 1998. PMID: 9658487 Review. Spanish.
-
Deficiency of the 50 kDa dystrophin-associated-glycoprotein (adhalin) in an Indian autosomal recessive limb girdle muscular dystrophy patient : immunochemical analysis and clinical aspects.Neurol India. 2001 Mar;49(1):19-24. Neurol India. 2001. PMID: 11303236
-
[Dystrophin and dystrophin-associated proteins. Their evaluation at the neuromuscular pathology laboratory].Rev Neurol. 1999 Jan 16-31;28(2):154-8. Rev Neurol. 1999. PMID: 10101784 Review. Spanish.
Cited by
-
Comparison of dysferlin expression in human skeletal muscle with that in monocytes for the diagnosis of dysferlin myopathy.PLoS One. 2011;6(12):e29061. doi: 10.1371/journal.pone.0029061. Epub 2011 Dec 16. PLoS One. 2011. PMID: 22194990 Free PMC article.
-
Antisense Oligonucleotide Treatment in a Humanized Mouse Model of Duchenne Muscular Dystrophy and Highly Sensitive Detection of Dystrophin Using Western Blotting.Methods Mol Biol. 2021;2224:203-214. doi: 10.1007/978-1-0716-1008-4_15. Methods Mol Biol. 2021. PMID: 33606217
-
Titin mutation segregates with hereditary myopathy with early respiratory failure.Brain. 2012 Jun;135(Pt 6):1695-713. doi: 10.1093/brain/aws102. Epub 2012 May 9. Brain. 2012. PMID: 22577215 Free PMC article.
-
Novel deletion of lysine 7 expands the clinical, histopathological and genetic spectrum of TPM2-related myopathies.Brain. 2013 Feb;136(Pt 2):508-21. doi: 10.1093/brain/aws344. Brain. 2013. PMID: 23413262 Free PMC article.
-
Intraarterial injection of muscle-derived CD34(+)Sca-1(+) stem cells restores dystrophin in mdx mice.J Cell Biol. 2001 Jan 22;152(2):335-48. doi: 10.1083/jcb.152.2.335. J Cell Biol. 2001. PMID: 11266450 Free PMC article.
References
-
- Bushby KMD, Beckmann JS: The limb-girdle muscular dystrophies - proposal for a new nomenclature (30th and 31st ENMC Workshops Report). Neuromuscul Disord 1995, 5:337-343 - PubMed
-
- Anderson LVB: Optimised protein diagnosis in the autosomal recessive limb-girdle muscular dystrophies. Neuromuscul Disord 1996, 6:443-446 - PubMed
-
- Tan E, Topaloglu H, Sewry C, Zorlu Y, Naom I, Erdem S, D’Alessandro M, Muntoni F, Dubowitz V: Late onset muscular dystrophy with cerebral white matter changes due to partial merosin deficiency. Neuromuscul Disord 1997, 7:85-89 - PubMed
-
- Muntoni F, Lichtarowicz-Krynska EJ, Sewry CA, Manilal S, Recan D, Llense S, Taylor J, Morris GE, Dubowitz V: Early presentation of X-linked Emery-Dreifuss muscular dystrophy resembling limb-girdle muscular dystrophy. Neuromuscul Disord 1998, 8:72-76 - PubMed
-
- Taylor J, Muntoni F, Robb S, Dubowitz V, Sewry C: Early onset autosomal dominant myopathy with rigidity of the spine: a possible role for laminin β1? Neuromuscul Disord 1997, 7:211-216 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

