Clinical and Genetic Characteristics of Congenital Hyperinsulinism in Norway: A Nationwide Cohort Study
- PMID: 38963811
- PMCID: PMC11747666
- DOI: 10.1210/clinem/dgae459
Clinical and Genetic Characteristics of Congenital Hyperinsulinism in Norway: A Nationwide Cohort Study
Abstract
Purpose: Congenital hyperinsulinism (CHI) is a rare, monogenic disease characterized by excessive insulin secretion. We aimed to evaluate all probands with suspected CHI in Norway registered over the past 2 decades.
Methods: The study included 98 probands. Clinical data were cumulated from medical records. All probands were screened for variants in the genes ABCC8 and KCNJ11. Other CHI-related genes were Sanger-sequenced as indicated by the patients' phenotype (n = 75) or analyzed by next-generation sequencing employing a panel of 30 CHI-related genes (n = 23).
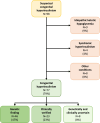
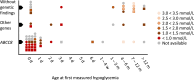
Results: Twenty-one probands (21%) received a diagnosis other than CHI, the most common being idiopathic ketotic hypoglycemia (9%) or syndromic hyperinsulinism (4%). In the final cohort of 77 CHI probands, genetic findings were revealed in 46 (60%). ABCC8 variants were most common (n= 40), and 5 novel variants were identified. One proband harbored both the pathogenic GCK variant p.(Ala456Val) and the ABCC8 variant p.(Gly505Cys). Although most ABCC8 variants caused immediate disease onset with severe hypoglycemia and were diazoxide-unresponsive, 8 probands had a heterozygous, apparently dominant variant with milder phenotype. Two probands had pathogenic variants in GLUD1, whereas variants in HADH, HNF4A, KCNJ11, and HK1 were identified in 1 proband each, the latter being noncoding. Neurologic sequelae were reported in 53% of the CHI probands. Of nonsurgically treated probands, 43% had spontaneous resolution. The minimum birth prevalence of CHI in Norway is 1:19,400 live births.
Main conclusion: Individuals with disease-causing ABCC8 variants dominated our cohort. Patients with known genetic etiology had earlier and more severe disease onset than genetically unsolved patients.
Keywords: genetic testing; hyperinsulinemic hypoglycemia; insulin dysregulation; monogenic disease; neurologic sequela; persistent hypoglycemia.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society.
Figures

Similar articles
-
Dominantly acting ABCC8 mutations in patients with medically unresponsive hyperinsulinaemic hypoglycaemia.Clin Genet. 2011 Jun;79(6):582-7. doi: 10.1111/j.1399-0004.2010.01476.x. Clin Genet. 2011. PMID: 20573158 Free PMC article.
-
Congenital hyperinsulinism in the Ukraine: a 10-year national study.Front Endocrinol (Lausanne). 2024 Dec 17;15:1497579. doi: 10.3389/fendo.2024.1497579. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39741883 Free PMC article.
-
Falls prevention interventions for community-dwelling older adults: systematic review and meta-analysis of benefits, harms, and patient values and preferences.Syst Rev. 2024 Nov 26;13(1):289. doi: 10.1186/s13643-024-02681-3. Syst Rev. 2024. PMID: 39593159 Free PMC article.
-
Adenosine Deaminase Deficiency.2006 Oct 3 [updated 2024 Mar 7]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2006 Oct 3 [updated 2024 Mar 7]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 20301656 Free Books & Documents. Review.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article. Review.
References
-
- Thornton PS, Stanley CA, De Leon DD. Congenital hyperinsulinism: an historical perspective. Horm Res Paediatr. 2022;95(6):631‐637. - PubMed
-
- Vora S, Chandran S, Rajadurai VS, Hussain K. Hyperinsulinemic hypoglycemia in infancy: current concepts in diagnosis and management. Indian Pediatr. 2015;52(12):1051‐1059. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

