Comparative Efficacy and Safety of Janus Kinase Inhibitors Used in Alopecia Areata: A Systematic Review and Meta-analysis
- PMID: 36695751
- PMCID: PMC10391778
- DOI: 10.2340/actadv.v103.4536
Comparative Efficacy and Safety of Janus Kinase Inhibitors Used in Alopecia Areata: A Systematic Review and Meta-analysis
Abstract
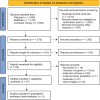
The aim of this study was to compare the efficacy and safety of treatment with Janus kinase inhibitors for alopecia areata, measured by change in Severity of Alopecia Tool (SALT) score. A systematic review following Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines was performed using Medline, EMBASE and Cochrane library. All studies investigating the efficacy of treatments for alopecia areata were included. Primary outcomes were the proportion of patients with alopecia areata achieving 30%, 50%, 75%, 90% and 100% improvement in SALT score after treatment with a Janus kinase inhibitor. A meta-analysis was performed including all randomized controlled trials investigating Janus kinase inhibitors. A total of 37 studies matched the inclusion criteria and were included. Meta-analysis was performed based on 5 randomized studies. Regarding patients with alopecia areata defined as ≥ 50% scalp hair loss, baricitinib 4 mg once daily demonstrated the highest efficacy. However, among patients with alopecia areata defined as a SALT score ≥ 50, oral deuruxolitinib 12 mg twice daily demonstrated the highest efficacy. Deuruxolitinib and baricitinib appear to be promising drugs for the treatment of alopecia areata. However, the response depends on the dosage of the drug. More randomized trials, with identical inclusion criteria and dose and duration of treatment, are required to confirm these findings.
Conflict of interest statement
MASH reported receiving grants from Leo Foundation. GBEJ reported receiving grants from AbbVie, LEO Foundation, Afyx, InflaRx, Janssen-Cilag, Novartis, UCB, CSL Behring, Regeneron, Sanofi, Boehringer Ingelheim, Union Therapeutics, and Toosonix and personal fees from Coloplast, Chemocentryx, LEO Pharma, Incyte, Kymera, and VielaBio. KSI has been part of advisory boards and received personal fees from Astra Zeneca, Leo Pharma; Sanofi Genzymes and Eli Lilly. MASH, GBEJ and KSI declare that none of the mentioned conflicts of interest had any influence to the content of this manuscript.
Figures
Comment in
-
Diese JAK-Inhibitoren helfen gegen Alopecia areata.MMW Fortschr Med. 2023 Feb;165(3):17. doi: 10.1007/s15006-023-2371-8. MMW Fortschr Med. 2023. PMID: 36759454 German. No abstract available.
Similar articles
-
Efficacy and safety of the oral Janus kinase inhibitor baricitinib in the treatment of adults with alopecia areata: Phase 2 results from a randomized controlled study.J Am Acad Dermatol. 2021 Oct;85(4):847-853. doi: 10.1016/j.jaad.2021.05.050. Epub 2021 Jun 16. J Am Acad Dermatol. 2021. PMID: 34090959 Clinical Trial.
-
Efficacy and safety of deuruxolitinib, an oral selective Janus kinase inhibitor, in adults with alopecia areata: Results from the Phase 3 randomized, controlled trial (THRIVE-AA1).J Am Acad Dermatol. 2024 Nov;91(5):880-888. doi: 10.1016/j.jaad.2024.06.097. Epub 2024 Jul 23. J Am Acad Dermatol. 2024. PMID: 39053611 Clinical Trial.
-
Safety of Janus Kinase inhibitors in Patients with Alopecia Areata: A Systematic Review.Clin Drug Investig. 2023 May;43(5):325-334. doi: 10.1007/s40261-023-01260-z. Epub 2023 May 3. Clin Drug Investig. 2023. PMID: 37138134 Free PMC article.
-
Exploring Janus kinase inhibitors for alopecia areata: a comprehensive review.Ital J Dermatol Venerol. 2024 Aug;159(4):380-389. doi: 10.23736/S2784-8671.24.07894-0. Epub 2024 May 23. Ital J Dermatol Venerol. 2024. PMID: 38780910 Review.
-
Janus kinase inhibitors for alopecia areata: A narrative review.Indian J Dermatol Venereol Leprol. 2023 Nov-Dec;89(6):799-806. doi: 10.25259/IJDVL_1093_2022. Indian J Dermatol Venereol Leprol. 2023. PMID: 37436019 Review.
Cited by
-
Research trends in alopecia areata: a cross-sectional bibliometric analysis of the top cited studies.Arch Dermatol Res. 2024 May 25;316(6):234. doi: 10.1007/s00403-024-03092-z. Arch Dermatol Res. 2024. PMID: 38795240
-
Patients' experiences of primary healthcare and dermatology provision for alopecia.Skin Health Dis. 2023 Dec 10;4(2):e324. doi: 10.1002/ski2.324. eCollection 2024 Apr. Skin Health Dis. 2023. PMID: 38577057 Free PMC article.
-
Establishing the financial burden of alopecia areata and its predictors.Skin Health Dis. 2023 Oct 15;4(1):e301. doi: 10.1002/ski2.301. eCollection 2024 Feb. Skin Health Dis. 2023. PMID: 38312246 Free PMC article.
-
Comparative efficacy and safety of JAK inhibitors in the treatment of moderate-to-severe alopecia areata: a systematic review and network meta-analysis.Front Pharmacol. 2024 Apr 10;15:1372810. doi: 10.3389/fphar.2024.1372810. eCollection 2024. Front Pharmacol. 2024. PMID: 38659584 Free PMC article.
-
Janus Kinase Inhibitors for Alopecia Areata: A Systematic Review and Meta-Analysis.JAMA Netw Open. 2023 Jun 1;6(6):e2320351. doi: 10.1001/jamanetworkopen.2023.20351. JAMA Netw Open. 2023. PMID: 37368402 Free PMC article.
References
-
- Yang S, Yang J, Liu JB, Wang HY, Yang Q, Gao M, et al. . The genetic epidemiology of alopecia areata in China. Br J Dermatol 2004; 151: 16–23. - PubMed
-
- Guzmán-Sánchez DA, Villanueva-Quintero GD, Alfaro Alfaro N, McMichael A. A clinical study of alopecia areata in Mexico. Int J Dermatol 2007; 46: 1308–1310. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources