How to conduct a systematic review and meta-analysis of prognostic model studies
- PMID: 35934199
- PMCID: PMC9351211
- DOI: 10.1016/j.cmi.2022.07.019
How to conduct a systematic review and meta-analysis of prognostic model studies
Abstract
Background: Prognostic models are typically developed to estimate the risk that an individual in a particular health state will develop a particular health outcome, to support (shared) decision making. Systematic reviews of prognostic model studies can help identify prognostic models that need to further be validated or are ready to be implemented in healthcare.
Objectives: To provide a step-by-step guidance on how to conduct and read a systematic review of prognostic model studies and to provide an overview of methodology and guidance available for every step of the review progress.
Sources: Published, peer-reviewed guidance articles.
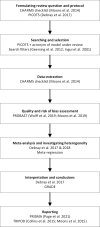
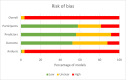
Content: We describe the following steps for conducting a systematic review of prognosis studies: 1) Developing the review question using the Population, Index model, Comparator model, Outcome(s), Timing, Setting format, 2) Searching and selection of articles, 3) Data extraction using the Critical Appraisal and Data Extraction for Systematic Reviews of Prediction Modelling Studies (CHARMS) checklist, 4) Quality and risk of bias assessment using the Prediction model Risk Of Bias ASsessment (PROBAST) tool, 5) Analysing data and undertaking quantitative meta-analysis, and 6) Presenting summary of findings, interpreting results, and drawing conclusions. Guidance for each step is described and illustrated using a case study on prognostic models for patients with COVID-19.
Implications: Guidance for conducting a systematic review of prognosis studies is available, but the implications of these reviews for clinical practice and further research highly depend on complete reporting of primary studies.
Keywords: Meta-analysis; Prediction model; Prognosis; Prognostic model; Systematic review.
Copyright © 2022 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Figures

Similar articles
-
Falls prevention interventions for community-dwelling older adults: systematic review and meta-analysis of benefits, harms, and patient values and preferences.Syst Rev. 2024 Nov 26;13(1):289. doi: 10.1186/s13643-024-02681-3. Syst Rev. 2024. PMID: 39593159 Free PMC article.
-
Development and validation of prediction models for fetal growth restriction and birthweight: an individual participant data meta-analysis.Health Technol Assess. 2024 Aug;28(47):1-119. doi: 10.3310/DABW4814. Health Technol Assess. 2024. PMID: 39252507 Free PMC article.
-
Genedrive kit for detecting single nucleotide polymorphism m.1555A>G in neonates and their mothers: a systematic review and cost-effectiveness analysis.Health Technol Assess. 2024 Oct;28(75):1-75. doi: 10.3310/TGAC4201. Health Technol Assess. 2024. PMID: 39487741 Free PMC article.
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
-
Continuing education meetings and workshops: effects on professional practice and healthcare outcomes.Cochrane Database Syst Rev. 2021 Sep 15;9(9):CD003030. doi: 10.1002/14651858.CD003030.pub3. Cochrane Database Syst Rev. 2021. PMID: 34523128 Free PMC article. Review.
Cited by
-
Developing clinical prediction models: a step-by-step guide.BMJ. 2024 Sep 3;386:e078276. doi: 10.1136/bmj-2023-078276. BMJ. 2024. PMID: 39227063 Free PMC article.
-
Value-Based Healthcare Delivery: A Scoping Review.Int J Environ Res Public Health. 2024 Jan 25;21(2):134. doi: 10.3390/ijerph21020134. Int J Environ Res Public Health. 2024. PMID: 38397625 Free PMC article.
-
Predicting Extended-Spectrum Beta-Lactamase and Carbapenem Resistance in Enterobacteriaceae Bacteremia: A Diagnostic Model Systematic Review and Meta-Analysis.Antibiotics (Basel). 2023 Sep 17;12(9):1452. doi: 10.3390/antibiotics12091452. Antibiotics (Basel). 2023. PMID: 37760748 Free PMC article. Review.
-
Clinical prognostic models in children with sepsis in low- and middle-income countries: a systematic review and meta-analysis.Front Pediatr. 2024 Oct 17;12:1463986. doi: 10.3389/fped.2024.1463986. eCollection 2024. Front Pediatr. 2024. PMID: 39483529 Free PMC article.
-
Predictive models of Alzheimer's disease dementia risk in older adults with mild cognitive impairment: a systematic review and critical appraisal.BMC Geriatr. 2024 Jun 19;24(1):531. doi: 10.1186/s12877-024-05044-8. BMC Geriatr. 2024. PMID: 38898411 Free PMC article.
References
-
- Moons K.G., Royston P., Vergouwe Y., Grobbee D.E., Altman D.G. Prognosis and prognostic research: what, why, and how? BMJ. 2009;338:b375. - PubMed
-
- Moons K.G., Kengne A.P., Woodward M., Royston P., Vergouwe Y., Altman D.G., et al. Risk prediction models: I. Development, internal validation, and assessing the incremental value of a new (bio)marker. Heart. 2012;98:683–690. - PubMed
-
- Steyerberg E. Springer Science & Business Media; Berlin, Germany: 2008. Clinical prediction models: a practical approach to development, validation, and updating.
-
- Riley R.D., van der Windt D., Croft P., Moons K.G. Oxford University Press; Oxford, UK: 2019. Prognosis research in healthcare: concepts, methods, and impact.
MeSH terms
LinkOut - more resources
Full Text Sources

