Metyrapone Versus Osilodrostat in the Short-Term Therapy of Endogenous Cushing's Syndrome: Results From a Single Center Cohort Study
- PMID: 35769081
- PMCID: PMC9235400
- DOI: 10.3389/fendo.2022.903545
Metyrapone Versus Osilodrostat in the Short-Term Therapy of Endogenous Cushing's Syndrome: Results From a Single Center Cohort Study
Abstract
Background: Although surgery is considered the first-line treatment for patients with endogenous Cushing's syndrome (CS), medical therapy is often required to control severe hypercortisolism. Metyrapone and osilodrostat are both steroidogenic inhibitors targeting the 11β-hydroxylase, however, their therapeutic effectiveness has not yet been directly compared. This study aimed to evaluate metyrapone and osilodrostat in the short-term therapy of CS.
Methods: Retrospective analysis of patients with endogenous CS treated with metyrapone or osilodrostat as monotherapy for at least 4 weeks. Main outcome measures were serum cortisol and 24h urinary free cortisol (UFC) at baseline (T0) and after 2 (T1), 4 (T2), and 12 weeks (T3) of therapy.
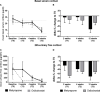
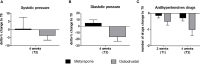
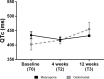
Results: 16 patients with endogenous CS were identified (pituitary n=7, adrenal n=4, ectopic CS n=5). Each 8 patients were treated with metyrapone and osilodrostat. Despite heterogeneity, both groups showed comparable mean UFC levels at T0 (metyrapone: 758 µg/24h vs osilodrostat: 817 µg/24h; p=0.93). From T0 to T1, the decrease of UFC was less pronounced under metyrapone than osilodrostat (-21.3% vs -68.4%; median daily drug dose: 1000 mg vs 4 mg). This tendency persisted at T2 (-37.3% vs -50.1%; median drug dose: 1250 mg vs 6 mg) while at T3 a decrease in UFC from T0 was more pronounced in the metyrapone group (-71.5% vs -51.5%; median dose 1250 mg vs 7 mg). Under osilodrostat, a QTc-interval prolongation was identified at T3 (mean 432 ms vs 455 ms). From T0 to T2, the number of antihypertensive drugs remained comparable under metyrapone and decreased under osilodrostat (n= -0.3 vs n= -1.0).
Conclusion: Although both drugs show comparable therapeutic efficacy, osilodrostat seems to reduce cortisol levels and to control blood pressure faster.
Keywords: Cushing’s syndrome; blood pressure; efficacy; hypercortisolism; isturisa; medical therapy; metyrapone; osilodrostat.
Copyright © 2022 Detomas, Altieri, Deutschbein, Fassnacht and Dischinger.
Conflict of interest statement
UD received honoraria from Recordati Rare Diseases for scientific board activities. TD received honoraria from Recordati Rare Diseases and HRA Pharma for scientific board activities. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Clinical Utility of Osilodrostat in Cushing's Disease: Review of Currently Available Literature.Drug Des Devel Ther. 2023 Apr 27;17:1303-1312. doi: 10.2147/DDDT.S315359. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 37143705 Free PMC article. Review.
-
Differences in the spectrum of steroidogenic enzyme inhibition between Osilodrostat and Metyrapone in ACTH-dependent Cushing syndrome patients.Eur J Endocrinol. 2022 Jul 4;187(2):315-322. doi: 10.1530/EJE-22-0208. Print 2022 Aug 1. Eur J Endocrinol. 2022. PMID: 35699971
-
Efficacy and safety of osilodrostat in patients with Cushing's disease (LINC 3): a multicentre phase III study with a double-blind, randomised withdrawal phase.Lancet Diabetes Endocrinol. 2020 Sep;8(9):748-761. doi: 10.1016/S2213-8587(20)30240-0. Epub 2020 Jul 27. Lancet Diabetes Endocrinol. 2020. PMID: 32730798 Clinical Trial.
-
Effectiveness of Metyrapone in Treating Cushing's Syndrome: A Retrospective Multicenter Study in 195 Patients.J Clin Endocrinol Metab. 2015 Nov;100(11):4146-54. doi: 10.1210/jc.2015-2616. Epub 2015 Sep 9. J Clin Endocrinol Metab. 2015. PMID: 26353009 Free PMC article.
-
Updates in adrenal steroidogenesis inhibitors for Cushing's syndrome - A practical guide.Best Pract Res Clin Endocrinol Metab. 2021 Jan;35(1):101490. doi: 10.1016/j.beem.2021.101490. Epub 2021 Feb 6. Best Pract Res Clin Endocrinol Metab. 2021. PMID: 33707082 Review.
Cited by
-
Response to Osilodrostat Therapy in Adrenal Cushing's Syndrome.Drug Healthc Patient Saf. 2024 Apr 8;16:35-42. doi: 10.2147/DHPS.S453105. eCollection 2024. Drug Healthc Patient Saf. 2024. PMID: 38616817 Free PMC article.
-
Erythropoiesis in Cushing syndrome: sex-related and subtype-specific differences. Results from a monocentric study.J Endocrinol Invest. 2024 Jan;47(1):101-113. doi: 10.1007/s40618-023-02128-x. Epub 2023 Jun 14. J Endocrinol Invest. 2024. PMID: 37314685 Free PMC article.
-
Individualized medical treatment options in Cushing disease.Front Endocrinol (Lausanne). 2022 Dec 2;13:1060884. doi: 10.3389/fendo.2022.1060884. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36531477 Free PMC article. Review.
-
Clinical Utility of Osilodrostat in Cushing's Disease: Review of Currently Available Literature.Drug Des Devel Ther. 2023 Apr 27;17:1303-1312. doi: 10.2147/DDDT.S315359. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 37143705 Free PMC article. Review.
-
Osilodrostat: A Novel Potent Inhibitor of 11-Beta-Hydroxylase for the Treatment of Cushing's Syndrome.touchREV Endocrinol. 2024 Apr;20(1):43-51. doi: 10.17925/EE.2024.20.1.8. Epub 2023 Dec 11. touchREV Endocrinol. 2024. PMID: 38812665 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

