Are Bacterial Volatile Compounds Poisonous Odors to a Fungal Pathogen Botrytis cinerea, Alarm Signals to Arabidopsis Seedlings for Eliciting Induced Resistance, or Both?
- PMID: 26941721
- PMCID: PMC4763075
- DOI: 10.3389/fmicb.2016.00196
Are Bacterial Volatile Compounds Poisonous Odors to a Fungal Pathogen Botrytis cinerea, Alarm Signals to Arabidopsis Seedlings for Eliciting Induced Resistance, or Both?
Abstract
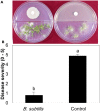
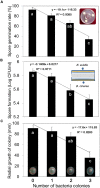
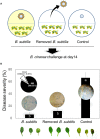
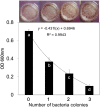
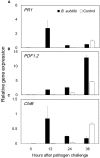
Biological control (biocontrol) agents act on plants via numerous mechanisms, and can be used to protect plants from pathogens. Biocontrol agents can act directly as pathogen antagonists or competitors or indirectly to promote plant induced systemic resistance (ISR). Whether a biocontrol agent acts directly or indirectly depends on the specific strain and the pathosystem type. We reported previously that bacterial volatile organic compounds (VOCs) are determinants for eliciting plant ISR. Emerging data suggest that bacterial VOCs also can directly inhibit fungal and plant growth. The aim of the current study was to differentiate direct and indirect mechanisms of bacterial VOC effects against Botrytis cinerea infection of Arabidopsis. Volatile emissions from Bacillus subtilis GB03 successfully protected Arabidopsis seedlings against B. cinerea. First, we investigated the direct effects of bacterial VOCs on symptom development and different phenological stages of B. cinerea including spore germination, mycelial attachment to the leaf surface, mycelial growth, and sporulation in vitro and in planta. Volatile emissions inhibited hyphal growth in a dose-dependent manner in vitro, and interfered with fungal attachment on the hydrophobic leaf surface. Second, the optimized bacterial concentration that did not directly inhibit fungal growth successfully protected Arabidopsis from fungal infection, which indicates that bacterial VOC-elicited plant ISR has a more important role in biocontrol than direct inhibition of fungal growth on Arabidopsis. We performed qRT-PCR to investigate the priming of the defense-related genes PR1, PDF1.2, and ChiB at 0, 12, 24, and 36 h post-infection and 14 days after the start of plant exposure to bacterial VOCs. The results indicate that bacterial VOCs potentiate expression of PR1 and PDF1.2 but not ChiB, which stimulates SA- and JA-dependent signaling pathways in plant ISR and protects plants against pathogen colonization. This study provides new evidence for bacterial VOC-elicited plant ISR that protects Arabidopsis plants from infection by the necrotrophic fungus B. cinerea. Our work reveals that bacterial VOCs primarily act via an indirect mechanism to elicit plant ISR, and have a major role in biocontrol against fungal pathogens.
Keywords: bacterial volatile organic compounds; biofilm formation; induced systemic resistance; leaf surface attachment; phytohormones; plant growth-promoting rhizobacteria.
Figures
Similar articles
-
Making healthier or killing enemies? Bacterial volatile-elicited plant immunity plays major role upon protection of Arabidopsis than the direct pathogen inhibition.Commun Integr Biol. 2016 Jul 12;9(4):e1197445. doi: 10.1080/19420889.2016.1197445. eCollection 2016 Jul-Aug. Commun Integr Biol. 2016. PMID: 27574539 Free PMC article.
-
Induced resistance by a long-chain bacterial volatile: elicitation of plant systemic defense by a C13 volatile produced by Paenibacillus polymyxa.PLoS One. 2012;7(11):e48744. doi: 10.1371/journal.pone.0048744. Epub 2012 Nov 28. PLoS One. 2012. PMID: 23209558 Free PMC article.
-
Bacillus proteolyticus OSUB18 triggers induced systemic resistance against bacterial and fungal pathogens in Arabidopsis.Front Plant Sci. 2023 Jan 23;14:1078100. doi: 10.3389/fpls.2023.1078100. eCollection 2023. Front Plant Sci. 2023. PMID: 36755698 Free PMC article.
-
Bacillus subtilis: A plant-growth promoting rhizobacterium that also impacts biotic stress.Saudi J Biol Sci. 2019 Sep;26(6):1291-1297. doi: 10.1016/j.sjbs.2019.05.004. Epub 2019 May 20. Saudi J Biol Sci. 2019. PMID: 31516360 Free PMC article. Review.
-
Interactions of Bacillus spp. and plants--with special reference to induced systemic resistance (ISR).Microbiol Res. 2009;164(5):493-513. doi: 10.1016/j.micres.2008.08.007. Epub 2008 Oct 8. Microbiol Res. 2009. PMID: 18845426 Review.
Cited by
-
C4 Bacterial Volatiles Improve Plant Health.Pathogens. 2021 May 31;10(6):682. doi: 10.3390/pathogens10060682. Pathogens. 2021. PMID: 34072921 Free PMC article. Review.
-
Priming Seeds with Indole and (Z)-3-Hexenyl Acetate Enhances Resistance Against Herbivores and Stimulates Growth.J Chem Ecol. 2022 Apr;48(4):441-454. doi: 10.1007/s10886-022-01359-1. Epub 2022 Apr 8. J Chem Ecol. 2022. PMID: 35394556
-
Pseudomonas aeruginosa isolate PM1 effectively controls virus infection and promotes growth in plants.Arch Microbiol. 2022 Jul 16;204(8):494. doi: 10.1007/s00203-022-03105-3. Arch Microbiol. 2022. PMID: 35841497
-
Microbiome-Mediated Strategies to Manage Major Soil-Borne Diseases of Tomato.Plants (Basel). 2024 Jan 25;13(3):364. doi: 10.3390/plants13030364. Plants (Basel). 2024. PMID: 38337897 Free PMC article. Review.
-
The Search for Quorum Sensing in Botrytis cinerea: Regulatory Activity of Its Extracts on Its Development.Plants (Basel). 2020 Jan 31;9(2):168. doi: 10.3390/plants9020168. Plants (Basel). 2020. PMID: 32023821 Free PMC article.
References
-
- Agrios G. N. (2004). Plant Pathology, 5th Edn. San Diego: Academic Press.
-
- Ann M. N., Cho Y. E., Ryu H. J., Kim H. T., Park K. S. (2013). Growth promotion of tobacco plant by 3-hydroxy-2-butanone from Bacillus vallismortis EXTN-1. Korean J. Pesti. Sci. 17 388–393. 10.7585/kjps.2013.17.4.388 - DOI
-
- Buensanteai N., Yuen G. Y., Prathuangwong S. (2009). Priming, signaling, and protein production associated with induced resistance by Bacillus amyloliquefaciens KPS46. World J. Microbiol. Biotechnol. 25 1275–1286. 10.1007/s11274-009-0014-6 - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

