RSPH3 Mutations Cause Primary Ciliary Dyskinesia with Central-Complex Defects and a Near Absence of Radial Spokes
- PMID: 26073779
- PMCID: PMC4571005
- DOI: 10.1016/j.ajhg.2015.05.004
RSPH3 Mutations Cause Primary Ciliary Dyskinesia with Central-Complex Defects and a Near Absence of Radial Spokes
Abstract
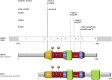
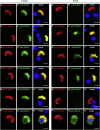
Primary ciliary dyskinesia (PCD) is a rare autosomal-recessive condition resulting from structural and/or functional defects of the axoneme in motile cilia and sperm flagella. The great majority of mutations identified so far involve genes whose defects result in dynein-arm anomalies. By contrast, PCD due to CC/RS defects (those in the central complex [CC] and radial spokes [RSs]), which might be difficult to diagnose, remains mostly unexplained. We identified non-ambiguous RSPH3 mutations in 5 of 48 independent families affected by CC/RS defects. RSPH3, whose ortholog in the flagellated alga Chlamydomonas reinhardtii encodes a RS-stalk protein, is mainly expressed in respiratory and testicular cells. Its protein product, which localizes within the cilia of respiratory epithelial cells, was undetectable in airway cells from an individual with RSPH3 mutations and in whom RSPH23 (a RS-neck protein) and RSPH1 and RSPH4A (RS-head proteins) were found to be still present within cilia. In the case of RSPH3 mutations, high-speed-videomicroscopy analyses revealed the coexistence of immotile cilia and motile cilia with movements of reduced amplitude. A striking feature of the ultrastructural phenotype associated with RSPH3 mutations is the near absence of detectable RSs in all cilia in combination with a variable proportion of cilia with CC defects. Overall, this study shows that RSPH3 mutations contribute to disease in more than 10% of PCD-affected individuals with CC/RS defects, thereby allowing an accurate diagnosis to be made in such cases. It also unveils the key role of RSPH3 in the proper building of RSs and the CC in humans.
Copyright © 2015 The American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
-
Defining the optimum strategy for identifying adults and children with coeliac disease: systematic review and economic modelling.Health Technol Assess. 2022 Oct;26(44):1-310. doi: 10.3310/ZUCE8371. Health Technol Assess. 2022. PMID: 36321689 Free PMC article.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article. Review.
-
Qualitative evidence synthesis informing our understanding of people's perceptions and experiences of targeted digital communication.Cochrane Database Syst Rev. 2019 Oct 23;10(10):ED000141. doi: 10.1002/14651858.ED000141. Cochrane Database Syst Rev. 2019. PMID: 31643081 Free PMC article.
-
Loss-of-function mutations in RSPH1 cause primary ciliary dyskinesia with central-complex and radial-spoke defects.Am J Hum Genet. 2013 Sep 5;93(3):561-70. doi: 10.1016/j.ajhg.2013.07.013. Epub 2013 Aug 29. Am J Hum Genet. 2013. PMID: 23993197 Free PMC article.
Cited by
-
The evolving spectrum of ciliopathies and respiratory disease.Curr Opin Pediatr. 2016 Jun;28(3):339-47. doi: 10.1097/MOP.0000000000000358. Curr Opin Pediatr. 2016. PMID: 27070443 Free PMC article. Review.
-
Novel RSPH4A Variants Associated With Primary Ciliary Dyskinesia-Related Infertility in Three Chinese Families.Front Genet. 2022 Jun 22;13:922287. doi: 10.3389/fgene.2022.922287. eCollection 2022. Front Genet. 2022. PMID: 35812741 Free PMC article.
-
Mapping the Most Common Founder Variant in RSPH9 That Causes Primary Ciliary Dyskinesia in Multiple Consanguineous Families of Bedouin Arabs.J Clin Med. 2023 Oct 13;12(20):6505. doi: 10.3390/jcm12206505. J Clin Med. 2023. PMID: 37892643 Free PMC article.
-
Primary Ciliary Dyskinesia (PCD): A genetic disorder of motile cilia.Transl Sci Rare Dis. 2019;4(1-2):51-75. doi: 10.3233/TRD-190036. Epub 2019 Jul 4. Transl Sci Rare Dis. 2019. PMID: 31572664 Free PMC article. No abstract available.
-
Emerging Genotype-Phenotype Relationships in Primary Ciliary Dyskinesia.Int J Mol Sci. 2021 Jul 31;22(15):8272. doi: 10.3390/ijms22158272. Int J Mol Sci. 2021. PMID: 34361034 Free PMC article. Review.
References
-
- Afzelius B.A. A human syndrome caused by immotile cilia. Science. 1976;193:317–319. - PubMed
-
- Afzelius B.A. The immotile-cilia syndrome: a microtubule-associated defect. CRC Crit. Rev. Biochem. 1985;19:63–87. - PubMed
-
- Satir P., Christensen S.T. Overview of structure and function of mammalian cilia. Annu. Rev. Physiol. 2007;69:377–400. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

