Platinum-induced neurotoxicity and preventive strategies: past, present, and future
- PMID: 25765877
- PMCID: PMC4391771
- DOI: 10.1634/theoncologist.2014-0044
Platinum-induced neurotoxicity and preventive strategies: past, present, and future
Abstract
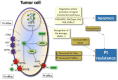
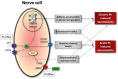
Neurotoxicity is a burdensome side effect of platinum-based chemotherapy that prevents administration of the full efficacious dosage and often leads to treatment withdrawal. Peripheral sensory neurotoxicity varies from paresthesia in fingers to ataxic gait, which might be transient or irreversible. Because the number of patients being treated with these neurotoxic agents is still increasing, the need for understanding the pathogenesis of this dramatic side effect is critical. Platinum derivatives, such as cisplatin and carboplatin, harm mainly peripheral nerves and dorsal root ganglia neurons, possibly because of progressive DNA-adduct accumulation and inhibition of DNA repair pathways (e.g., extracellular signal-regulated kinase 1/2, c-Jun N-terminal kinase/stress-activated protein kinase, and p38 mitogen-activated protein kinass), which finally mediate apoptosis. Oxaliplatin, with a completely different pharmacokinetic profile, may also alter calcium-sensitive voltage-gated sodium channel kinetics through a calcium ion immobilization by oxalate residue as a calcium chelator and cause acute neurotoxicity. Polymorphisms in several genes, such as voltage-gated sodium channel genes or genes affecting the activity of pivotal metal transporters (e.g., organic cation transporters, organic cation/carnitine transporters, and some metal transporters, such as the copper transporters, and multidrug resistance-associated proteins), can also influence drug neurotoxicity and treatment response. However, most pharmacogenetics studies need to be elucidated by robust evidence. There are supportive reports about the effectiveness of several neuroprotective agents (e.g., vitamin E, glutathione, amifostine, xaliproden, and venlafaxine), but dose adjustment and/or drug withdrawal seem to be the most frequently used methods in the management of platinum-induced peripheral neurotoxicity. To develop alternative options in the treatment of platinum-induced neuropathy, studies on in vitro models and appropriate trials planning should be integrated into the future design of neuroprotective strategies to find the best patient-oriented solution.
Keywords: Models; Neurotoxicity; Pathogenesis; Platinum; Polymorphism; Prevention.
©AlphaMed Press.
Conflict of interest statement
Disclosures of potential conflicts of interest may be found at the end of this article.
Figures
Similar articles
-
Platinum neurotoxicity: clinical profiles, experimental models and neuroprotective approaches.J Inorg Biochem. 1999 Oct;77(1-2):105-10. doi: 10.1016/s0162-0134(99)00135-x. J Inorg Biochem. 1999. PMID: 10626361 Review.
-
The role of oxidative stress and anti-oxidant treatment in platinum-induced peripheral neurotoxicity.Curr Cancer Drug Targets. 2010 Nov;10(7):670-82. doi: 10.2174/156800910793605820. Curr Cancer Drug Targets. 2010. PMID: 20578989 Review.
-
Interventions for preventing neuropathy caused by cisplatin and related compounds.Cochrane Database Syst Rev. 2014 Mar 31;2014(3):CD005228. doi: 10.1002/14651858.CD005228.pub4. Cochrane Database Syst Rev. 2014. PMID: 24687190 Free PMC article. Review.
-
[Oxaliplatin neurotoxicity].Bull Cancer. 2006 Feb 1;93 Suppl 1:S17-22. Bull Cancer. 2006. PMID: 16491518 Review. French.
-
Neuronal drug transporters in platinum drugs-induced peripheral neurotoxicity.Anticancer Res. 2014 Jan;34(1):483-6. Anticancer Res. 2014. PMID: 24403505 Review.
Cited by
-
Design of Targeted Nanostructured Coordination Polymers (NCPs) for Cancer Therapy.Molecules. 2020 Jul 29;25(15):3449. doi: 10.3390/molecules25153449. Molecules. 2020. PMID: 32751178 Free PMC article. Review.
-
Berberis lyceum root bark extract attenuates anticancer drugs induced neurotoxicityand cardiotoxicity in rats.Afr Health Sci. 2022 Sep;22(3):192-210. doi: 10.4314/ahs.v22i3.22. Afr Health Sci. 2022. PMID: 36910359 Free PMC article.
-
Association between Genetic Variants and Peripheral Neuropathy in Patients with NSCLC Treated with First-Line Platinum-Based Therapy.Genes (Basel). 2023 Jan 7;14(1):170. doi: 10.3390/genes14010170. Genes (Basel). 2023. PMID: 36672910 Free PMC article.
-
Heterodinuclear Ru-Pt Complexes Bridged with 2,3-Bis(pyridyl)pyrazinyl Ligands: Studies on Kinetics, Deoxyribonucleic Acid/Bovine Serum Albumin Binding and Cleavage, In Vitro Cytotoxicity, and In Vivo Toxicity on Zebrafish Embryo Activities.ACS Omega. 2022 Jul 21;7(30):26226-26245. doi: 10.1021/acsomega.2c01845. eCollection 2022 Aug 2. ACS Omega. 2022. PMID: 35936428 Free PMC article.
-
Chemotherapy-induced neuropathies-a growing problem for patients and health care providers.Brain Behav. 2016 Oct 26;7(1):e00558. doi: 10.1002/brb3.558. eCollection 2017 Jan. Brain Behav. 2016. PMID: 28127506 Free PMC article. Review.
References
-
- Fuertes MA, Castilla J, Alonso C, et al. Novel concepts in the development of platinum antitumor drugs. Curr Med Chem Anticancer Agents. 2002;2:539–551. - PubMed
-
- Boulikas T, Pantos A, Bellis E, et al. Designing platinum compounds in cancer: Structures and mechanisms. Cancer Ther. 2007;5:537–583.
-
- Ali I, Wani WA, Saleem K, et al. Platinum compounds: A hope for future cancer chemotherapy. Anticancer Agents Med Chem. 2013;13:296–306. - PubMed
-
- Argyriou AA, Bruna J, Marmiroli P, et al. Chemotherapy-induced peripheral neurotoxicity (CIPN): An update. Crit Rev Oncol Hematol. 2012;82:51–77. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

