LINC complexes form by binding of three KASH peptides to domain interfaces of trimeric SUN proteins
- PMID: 22632968
- PMCID: PMC3383001
- DOI: 10.1016/j.cell.2012.03.046
LINC complexes form by binding of three KASH peptides to domain interfaces of trimeric SUN proteins
Abstract
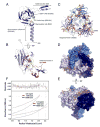
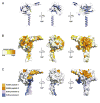
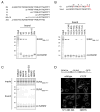
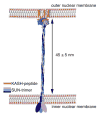
Linker of nucleoskeleton and cytoskeleton (LINC) complexes span the nuclear envelope and are composed of KASH and SUN proteins residing in the outer and inner nuclear membrane, respectively. LINC formation relies on direct binding of KASH and SUN in the perinuclear space. Thereby, molecular tethers are formed that can transmit forces for chromosome movements, nuclear migration, and anchorage. We present crystal structures of the human SUN2-KASH1/2 complex, the core of the LINC complex. The SUN2 domain is rigidly attached to a trimeric coiled coil that prepositions it to bind three KASH peptides. The peptides bind in three deep and expansive grooves formed between adjacent SUN domains, effectively acting as molecular glue. In addition, a disulfide between conserved cysteines on SUN and KASH covalently links both proteins. The structure provides the basis of LINC complex formation and suggests a model for how LINC complexes might arrange into higher-order clusters to enhance force-coupling.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures


Comment in
-
It takes KASH to hitch to the SUN.Cell. 2012 May 25;149(5):961-3. doi: 10.1016/j.cell.2012.05.004. Cell. 2012. PMID: 22632963 No abstract available.
Similar articles
-
Structural insights into SUN-KASH complexes across the nuclear envelope.Cell Res. 2012 Oct;22(10):1440-52. doi: 10.1038/cr.2012.126. Epub 2012 Sep 4. Cell Res. 2012. PMID: 22945352 Free PMC article.
-
Structure of Sad1-UNC84 homology (SUN) domain defines features of molecular bridge in nuclear envelope.J Biol Chem. 2012 Feb 17;287(8):5317-26. doi: 10.1074/jbc.M111.304543. Epub 2011 Dec 14. J Biol Chem. 2012. PMID: 22170055 Free PMC article.
-
Conserved SUN-KASH Interfaces Mediate LINC Complex-Dependent Nuclear Movement and Positioning.Curr Biol. 2018 Oct 8;28(19):3086-3097.e4. doi: 10.1016/j.cub.2018.08.001. Epub 2018 Sep 20. Curr Biol. 2018. PMID: 30245107 Free PMC article.
-
SUN/KASH interactions facilitate force transmission across the nuclear envelope.Nucleus. 2019 Dec;10(1):73-80. doi: 10.1080/19491034.2019.1595313. Nucleus. 2019. PMID: 30888237 Free PMC article. Review.
-
How plants LINC the SUN to KASH.Nucleus. 2013 May-Jun;4(3):206-15. doi: 10.4161/nucl.24088. Epub 2013 May 13. Nucleus. 2013. PMID: 23680964 Free PMC article. Review.
Cited by
-
A human infertility-associated KASH5 variant promotes mitochondrial localization.Sci Rep. 2021 May 12;11(1):10133. doi: 10.1038/s41598-021-89439-2. Sci Rep. 2021. PMID: 33980926 Free PMC article.
-
Effects of mutant lamins on nucleo-cytoskeletal coupling in Drosophila models of LMNA muscular dystrophy.Front Cell Dev Biol. 2022 Aug 31;10:934586. doi: 10.3389/fcell.2022.934586. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36120560 Free PMC article.
-
The Diverse Cellular Functions of Inner Nuclear Membrane Proteins.Cold Spring Harb Perspect Biol. 2021 Sep 1;13(9):a040477. doi: 10.1101/cshperspect.a040477. Cold Spring Harb Perspect Biol. 2021. PMID: 33753404 Free PMC article. Review.
-
The NAE Pathway: Autobahn to the Nucleus for Cell Surface Receptors.Cells. 2019 Aug 16;8(8):915. doi: 10.3390/cells8080915. Cells. 2019. PMID: 31426451 Free PMC article. Review.
-
The mammalian LINC complex component SUN1 regulates muscle regeneration by modulating drosha activity.Elife. 2019 Nov 5;8:e49485. doi: 10.7554/eLife.49485. Elife. 2019. PMID: 31686651 Free PMC article.
References
-
- Apel ED, Lewis RM, Grady RM, Sanes JR. Syne-1, a dystrophin- and Klarsicht-related protein associated with synaptic nuclei at the neuromuscular junction. J Biol Chem. 2000;275:31986–31995. - PubMed
-
- Appenzeller-Herzog C, Ellgaard L. The human PDI family: versatility packed into a single fold. Biochim Biophys Acta. 2008;1783:535–548. - PubMed
-
- Bianchet MA, Odom EW, Vasta GR, Amzel LM. A novel fucose recognition fold involved in innate immunity. Nat Struct Biol. 2002;9:628–634. - PubMed
-
- Burke B, Roux KJ. Nuclei take a position: managing nuclear location. Dev Cell. 2009;17:587–597. - PubMed
-
- Chikashige Y, Tsutsumi C, Yamane M, Okamasa K, Haraguchi T, Hiraoka Y. Meiotic proteins bqt1 and bqt2 tether telomeres to form the bouquet arrangement of chromosomes. Cell. 2006;125:59–69. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

